HeatPage
3
3
Slide 20
Convection
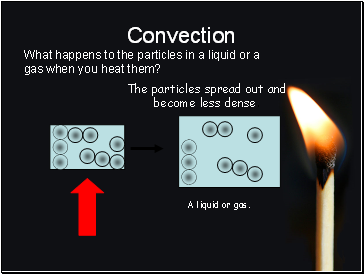
What happens to the particles in a liquid or a gas when you heat them?
The particles spread out and become less dense.
A liquid or gas.
Slide 21
Convection
It is the way in which particles in a GAS or LIQUID move upwards, carrying heat with them
Think about when you boil water, the bubbles move upwards
Or think of a gas heater in the room, the heat rises around the room
Slide 22
Convection
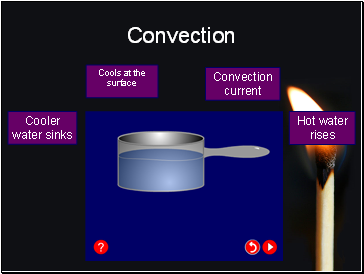
Hot water rises
Cooler water sinks
Convection current
Cools at the surface
Slide 23
Convection
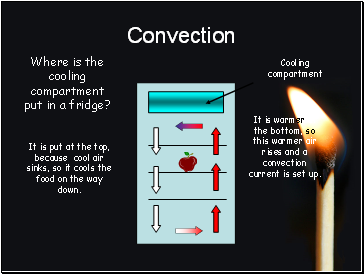
Where is the cooling compartment put in a fridge?
Cooling compartment
It is put at the top, because cool air sinks, so it cools the food on the way down.
It is warmer at the bottom, so this warmer air rises and a convection current is set up.
Slide 24
Should a radiator be called a radiator?
Slide 25
Convection questions
Why are boilers placed beneath hot water tanks in people’s homes?
Hot water rises.
So when the boiler heats the water, and the hot water rises, the water tank is filled with hot water.
Why does hot air rise and cold air sink?
Cool air is more dense than warm air, so the cool air ‘falls through’ the warm air.
Slide 26
Heat Vs Temperature
The temperature of an object tells us how HOT it is
Measured in degrees Celsius - °C
It is NOT the same as heat energy although the two quantities are related. e.g. a beaker of water at 60 °C is hotter than a bath of water at 40 °C BUT the bath contains more joules of heat energy
Slide 27
Heating and Cooling
If an object has become hotter, it means that it has gained heat energy.
If an object cools down, it means it has lost energy
Slide 28
Heating and Cooling cont…
Heat energy always moves from:
HOT object COOLER object
e.g. Cup of water at 20 °C in a room at 30°C - gains heat energy and heats up – its temperature rises
Cup of water at 20 °C in a room at 10°C loses heat energy and cools down – its temperature will fall.
Slide 29
Expansion/Contraction
Contents
- By the end of this chapter you will be able to
- What is Heat?
- Heat can be converted to other forms of energy
- How is heat produced?
- Heat Transfer
- Forms of Heat Transfer
- Radiation
- What colour should we paint radiators? Which colour is better to wear on a sunny day? black or white?
- Radiation – Think Pair-Share
- Radiation questions
- Conduction
- Conductors/Insulators
- Conductor or Insulator?
- Convection
- Should a radiator be called a radiator?
- Heat Vs Temperature
- Heating and Cooling
- Heating and Cooling cont…
- Expansion/Contraction
- Expansion V Contraction
- Revision
Last added presentations
- Newton’s law of universal gravitation
- Heat-Energy on the Move
- Sound
- Health Physics
- Newton’s laws of motion
- Sensory and Motor Mechanisms
- Resource Acquisition and Transport in Vascular Plants