Scientific MeasurementPage
2
2
The cylinder contains:
_ _ . _ mL
7
6
0
Slide 12
The Thermometer
Determine the temperature by reading the scale on the thermometer at eye level.
Read the temperature by using all certain digits and one uncertain digit.
Certain digits are determined from the calibration marks on the thermometer.
The uncertain digit (the last digit of the reading) is estimated.
On most thermometers encountered in a general chemistry lab, the tenths place is the uncertain digit.
Slide 13
Do not allow the tip to touch the walls or the bottom of the flask.
If the thermometer bulb touches the flask, the temperature of the glass will be measured instead of the temperature of the solution. Readings may be incorrect, particularly if the flask is on a hotplate or in an ice bath.
Slide 14
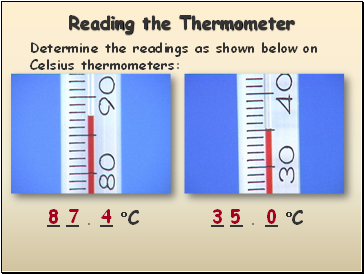
Reading the Thermometer
Determine the readings as shown below on Celsius thermometers:
_ _ . _ C
_ _ . _ C
8
7
4
3
5
0
Slide 15
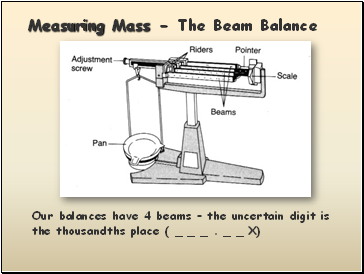
Measuring Mass - The Beam Balance
Our balances have 4 beams – the uncertain digit is the thousandths place ( _ _ _ . _ _ X)
Slide 16
Balance Rules
In order to protect the balances and ensure accurate results, a number of rules should be followed:
Always check that the balance is level and zeroed before using it.
Never weigh directly on the balance pan. Always use a piece of weighing paper to protect it.
Do not weigh hot or cold objects.
Clean up any spills around the balance immediately.
Slide 17
Mass and Significant Figures
Determine the mass by reading the riders on the beams at eye level.
Read the mass by using all certain digits and one uncertain digit.
The uncertain digit (the last digit of the reading) is estimated.
On our balances, the thousandths place is uncertain.
Slide 18
Determining Mass
1. Place object on pan
2. Move riders along beam, starting with the largest, until the pointer is at the zero mark
Slide 19
Check to see that the balance scale is at zero
Slide 20
Read Mass
_ _ _ . _ _ _
1
1
4
? ? ?
Slide 21
Contents
- Measuring
- Reading the Meniscus
- Try to avoid parallax errors.
- Graduated Cylinders
- Measuring Volume
- Use the graduations to find all certain digits
- Estimate the uncertain digit and take a reading
- 10 mL Graduate
- 25mL graduated cylinder
- 100mL graduated cylinder
- Self Test
- The Thermometer
- Reading the Thermometer
- Balance Rules
- Mass and Significant Figures
- Determining Mass
- Check to see that the balance scale is at zero
Last added presentations
- Space Radiation
- Mechanical, Electromagnetic, Electrical, Chemical and Thermal
- Sound
- Solar Thermal Energy
- Motion
- History of Modern Astronomy
- The Effects of Radiation on Living Things