CatalysisPage
1
1
Slide 1
Catalysis
Catalyst: A substance that speeds up a reaction without being consumed
Enzyme: A large molecule (usually a protein) that catalyzes biological reactions.
Homogeneous catalyst: Present in the same phase as the reacting molecules.
Heterogeneous catalyst: Present in a different phase than the reacting molecules.
Slide 2
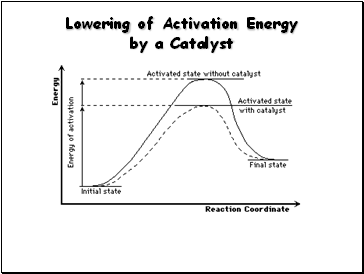
Lowering of Activation Energy by a Catalyst
Slide 3
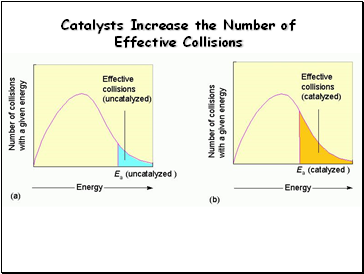
Catalysts Increase the Number of Effective Collisions
Slide 4
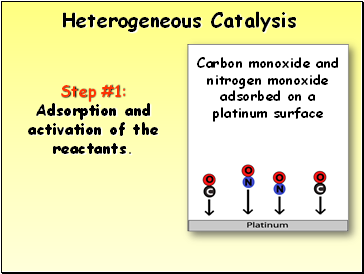
Heterogeneous Catalysis
Step #1: Adsorption and activation of the reactants.
Carbon monoxide and nitrogen monoxide adsorbed on a platinum surface
Slide 5
Heterogeneous Catalysis
Step #2:
Migration of the adsorbed reactants on the surface.
Carbon monoxide and nitrogen monoxide arranged prior to reacting
Slide 6
Heterogeneous Catalysis
Step #3:
Reaction of the adsorbed substances.
Carbon dioxide and nitrogen form from previous molecules
Slide 7
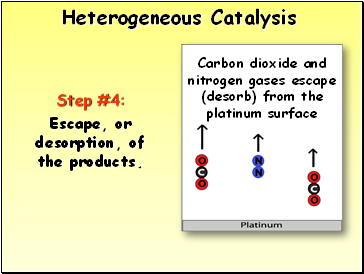
Heterogeneous Catalysis
Step #4:
Escape, or desorption, of the products.
Carbon dioxide and nitrogen gases escape (desorb) from the platinum surface
Contents
Last added presentations
- Madame Marie Curie
- Space Radiation
- Newton’s law of universal gravitation
- Health Physics
- Newton's Laws
- Radiation Safety and Operations
- Static and Kinetic Friction