The Sun and Its Ability to Create PowerPage
2
2
Nuclear physics deals with the structure of the nuclei of atoms.
The special theory of relativity deals with the behavior of things moving at close to the speed of light.
Slide 9
15.2.1 Converting Mass to Energy
Out of the special theory of relativity comes the most famous equation in science:
This equation tells us that mass (m) is just another form of energy (E)!
The c2 is the square of the speed of light.
1 gram of matter is equivalent to the energy obtained by burning 15,000 barrels of oil.
Slide 10
…but there are rules
We can’t simply convert atoms into energy.
We rearrange the protons and neutrons in nuclei to get a lower-mass configuration.
The difference between initial mass and final mass is converted to energy.
Chemical energy comes from rearranging atoms to configurations of lower energy (mass).
Nuclear energy comes from rearranging nuclei to configurations of lower mass (energy).
In each case, we get out the energy difference.
Slide 11
Slide 12
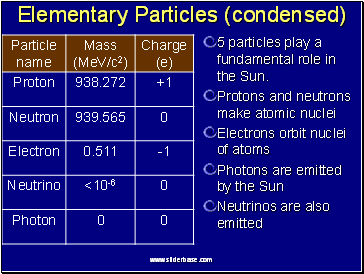
Elementary Particles (condensed)
5 particles play a fundamental role in the Sun.
Protons and neutrons make atomic nuclei
Electrons orbit nuclei of atoms
Photons are emitted by the Sun
Neutrinos are also emitted
Slide 13
15.2.3 The Atomic Nucleus
Two ways to rearrange nuclei and get energy:
Fission
produces energy by breaking up massive nuclei like Uranium into less massive nuclei like Barium and Krypton
A-bombs, nuclear reactors
needs Uranium 235, Plutonium 238
Problem: no Uranium or Plutonium on the Sun
Fusion
produces energy by combining light nuclei like Hydrogen to make more massive nuclei like Helium.
H-bomb, tokamak, internal confinement fusion
Sun has lots of Hydrogen!!
Slide 14
How Does Fusion Work?
Nuclear fusion:
a process by which two light nuclei combine to form a single larger nucleus.
However: nuclei are positively charged
Like charges repel
Two nuclei naturally repel each other and thus cannot fuse spontaneously
For fusion, electrical repulsion must be “overcome”
When two nuclei are very close the strong nuclear force takes over and holds them together.
How do two nuclei get close enough?
Slide 15
Fusion needs fast moving nuclei
Fast moving nuclei can overcome the repulsion - they get a running start.
Contents
- Happy Sun
- Why Does the Sun Shine?
- The Sun’s Energy Output
- Gravity Squeeze?
- Elementary Particles (condensed)
- Fusion needs fast moving nuclei
- Fusion Power on Earth
- Why a complicated chain?
- Gravity and Pressure
- Temperature and Pressure
- Hydrostatic Equilibrium
- Other Particles
- Neutrinos
- Neutrino Counting
Last added presentations
- Radiation Safety and Operations
- Sensory and Motor Mechanisms
- Sound
- Static and Kinetic Friction
- Sound
- Newton’s third law of motion
- Direct heat utilization of geothermal energy