Ch 9 Nuclear RadiationPage
2
2
Learning Check
To predict the product, simply remember that the mass number and atomic number are conserved
What is the identity of X?
Slide 14
Learning Check
Write the nuclear equation for the beta emitter Co-60.
Slide 15
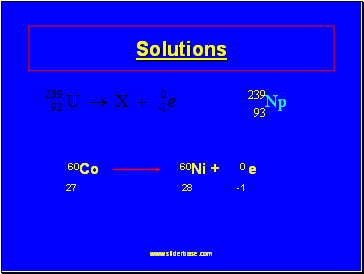
Solutions
60Co 60Ni + 0 e
27 28 -1
93
239
Np
Slide 16
Producing Radioactive Isotopes
Bombardment of atoms produces radioisotopes
= 60 = 60
59Co + 1n 56Mn + 4H e
27 0 25 2
= 27 = 27
cobalt neutron manganese alpha
atom radioisotope particle
Slide 17
Learning Check
What radioactive isotope is produced in the following bombardment of boron?
10B + 4He ? + 1n
5 2 0
Slide 18
Solution
What radioactive isotope is produced in the following bombardment of boron?
10B + 4He 13N + 1n
5 2 7 0
nitrogen
radioisotope
Slide 19
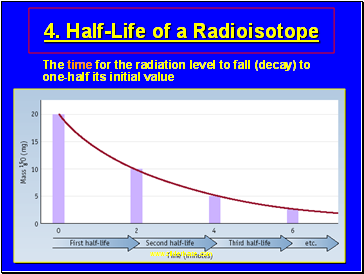
Half-Life of a Radioisotope
The time for the radiation level to fall (decay) to one-half its initial value
Slide 20
Examples of Half-Life
Isotope Half life
C-15 2.4 sec
Ra-224 3.6 days
Ra-223 12 days
I-125 60 days
C-14 5700 years
U-235 710 000 000 years
Slide 21
Learning Check
The half life of I-123 is 13 hr. How much of a 64 mg sample of I-123 is left after 26 hours?
Slide 22
Solution
t1/2 = 13 hrs
26 hours = 2 x t1/2
Amount initial = 64mg
Amount remaining = 64 mg x ½ x ½
= 16 mg
Slide 23
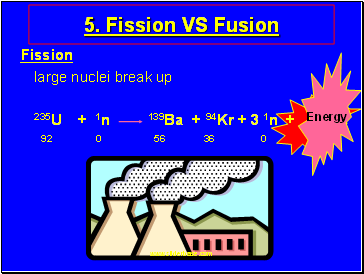
Fission VS Fusion
Fission
large nuclei break up
235U + 1n 139Ba + 94Kr + 3 1n +
92 0 56 36 0
Energy
Slide 24
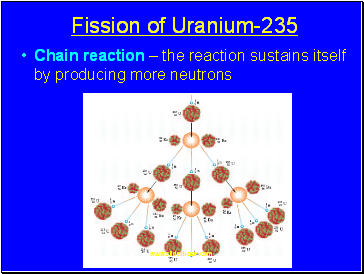
Fission of Uranium-235
Chain reaction – the reaction sustains itself by producing more neutrons
Slide 25
Nuclear Fusion
Fusion
small nuclei combine
2H + 3H 4He + 1n +
1 1 2 0
Occurs in the sun and other stars
Energy
Excessive heat can not be contained
Contents
- Ch 9 - Nuclear Radiation
- Nuclear Emissions
- Nuclear Equations
- Producing Radioactive Isotopes
- Half-Life of a Radioisotope
- Fission VS Fusion
- Uses & Effects
Last added presentations
- Newton's laws of motion
- History of Modern Astronomy
- Newton’s law of universal gravitation
- Radiation Safety and Operations
- Space Radiation
- Radioactivity and Nuclear Reactions
- Heat-Energy on the Move