Bohr Models and Lewis Dot DiagramsPage
1
1
Slide 1
Bohr Models and Lewis Dot Diagrams
Science 10
Slide 2
Niels Bohr
Slide 3
Bohr Models…
Niels Bohr explained the structure of the atom in his “Bohr models”. His model of the atom resembles a solar system. He came up with this model in 1913. Here is a typical Bohr model, what can you figure out from it?
Slide 4
Bohr models show the nucleus in the center with the # of Protons (p) and Neutrons (n) in it
Around the nucleus, it shows
the electrons in orbits or shells
Each orbit/shell can only hold a
certain # of electrons and then
it is full.
Slide 5
How many e- can each orbit hold?
1st orbit can hold 2 e-
2nd orbit can hold 8 e-
3rd orbit can hold 8 e-
4th orbit can hold 18 e-
You don’t need to know any higher orbits – we won’t be covering higher elements.
You can depict any element in a Bohr model!
Slide 6
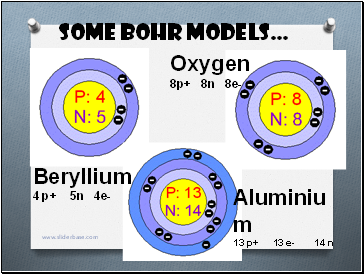
Some Bohr models…
Beryllium
4 p+ 5n 4e-
Oxygen
8p+ 8n 8e-
Aluminium
13 p+ 13 e- 14 n
Slide 7
So how do we draw a Bohr model of… say…Sodium?
Start with a nucleus, and put in how many protons and neutrons are in sodium (look at your periodic table)
Next place some orbits around it
P:11
N:12
P:11
N:12
Slide 8
Next figure out how many e- are in sodium (look at your periodic table). Place them in dots in the orbits
P:11
N:12
Remember!!
2 max in 1st orbit
8 max in 2nd orbit
8 max in 3rd orbit
18 max in 4th orbit
Sodium has 11 electrons !!
they are the same as the protons
Slide 9
Draw a Bohr Model for an Argon atom
How many neutrons and protons does it have?
How many electrons does it have?
Slide 10
Practice
Blank Periodic Table
Slide 11
Valence Electrons
Valence electrons and stability
Which group on the periodic table, do you notice, always has all its valence electron shell full? _
Every element wants to have a full outer shell – because then it is and never has to react.
Contents
- Bohr Models…
- How many e- can each orbit hold?
- Some Bohr models…
- So how do we draw a Bohr model of… say…Sodium?
- Draw a Bohr Model for an Argon atom
- Valence Electrons
- Lewis Dot Diagrams…
- Lewis Dot Structures
Last added presentations
- Solar Energy
- Waves & Sound
- Sound
- Newton’s law of universal gravitation
- Sensory and Motor Mechanisms
- Newton's Laws
- Simulation at NASA for the Space Radiation Effort