Electron Cloud ModelPage
1
1
Slide 1
Ch. 10 - Atomic Structure
II. Electron Cloud Model
(p.272-274)
Orbital
Energy Levels
Bohr Model Diagrams
Slide 2
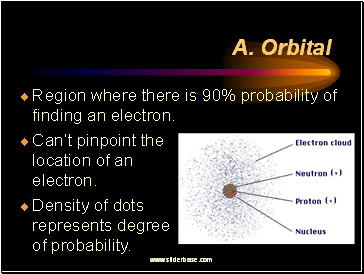
Orbital
Region where there is 90% probability of finding an electron.
Can’t pinpoint the location of an electron.
Density of dots represents degree of probability.
Slide 3
A. Orbital
Orbitals have different shapes.
Slide 4
Energy Levels
Electrons can only exist at certain energy levels.
Low energy levels are close to the nucleus.
Each energy level (n) can hold 2n2 electrons.
Slide 5
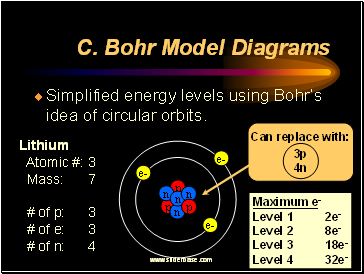
Bohr Model Diagrams
Simplified energy levels using Bohr’s idea of circular orbits.
e-
e-
Maximum e-
Level 1 2e-
Level 2 8e-
Level 3 18e-
Level 4 32e-
Lithium
Atomic #: 3
Mass: 7
# of p: 3
# of e: 3
# of n: 4
e-
Slide 6
C. Bohr Model Activity
Choose a number between 1 & 18.
Find your element by the atomic number you picked.
Draw a Bohr Model diagram for your element on your marker board.
Round off the mass listed on the table and subtract the atomic # to find the # of neutrons.
Abbreviate the # of ‘p’ and ‘n’ in the nucleus.
Have a partner check your drawing.
Repeat with a new element.
Contents
Last added presentations
- Newton’s laws of motion
- Waves & Sound
- Newton’s Laws of Motion
- Magnetic field uses sound waves to ignite sun's ring of fire
- The Effects of Radiation on Living Things
- Radiation
- Sound