The Building Blocks of Matter AtomsPage
1
1
Slide 1
The Building Blocks of Matter: Atoms
+
+
+
+
-
-
-
-
-
-
-
-
+
Slide 2
Matter
Anything that has mass and takes up space (volume)
Examples:
A brick has mass and takes up space
A desk has mass and takes up space
A pencil has mass and takes up space
Air has mass and takes up space
All of the above examples are considered matter because they have mass and take up space. Can you think of anything that would not be considered matter?
Slide 3
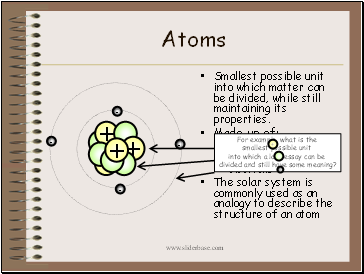
Atoms
Smallest possible unit into which matter can be divided, while still maintaining its properties.
Made up of:
protons
neutrons
electrons
The solar system is commonly used as an analogy to describe the structure of an atom
For example, what is the
smallest possible unit
into which a long essay can be
divided and still have some meaning?
+
-
+
+
+
-
-
-
-
+
Slide 4
Atoms are so small that…
it would take a stack of about 50,000 aluminum atoms to equal the thickness of a sheet of aluminum foil from your kitchen.
if you could enlarge a penny until it was as wide as the US, each of its atoms would be only about 3 cm in diameter – about the size of a ping-pong ball
a human hair is about 1 million carbon atoms wide.
a typical human cell contains roughly 1 trillion atoms.
a speck of dust might contain 3x1012 (3 trillion) atoms.
it would take you around 500 years to count the number of atoms in a grain of salt.
C-C-C-C-C-… + 999,995 more
1 trillion atoms
.
Is made of approximately 3 trillion atoms
Just one of these grains
Slide 5
Let’s Experiment
In order to try to gain an idea of how small an atom really is, you will complete the following activity.
Cut a strip of 11 in. paper in half.
Discard one half.
Cut the remaining piece in half.
Continue cutting and discarding the strips as many times as you can.
Make all cuts parallel to the first one. When the width gets longer than the length, you may cut off the excess, but that does not count as a cut.
Slide 6
Results
How many cuts were you able to make?
Do you think you could keep cutting the paper forever? Why or why not?
You would have to cut the paper in half around thirty-one (31) times to get to the size of any atom.
Contents
- Matter
- Atoms
- Atoms are so small that…
- Let’s Experiment
- Results
- Protons (+)
- Neutrons
- Electrons (-)
- Hydrogen (H) Atom
- Oxygen (O) Atom
- Sodium (Na) Atom
- The Atom’s “Center”
- Quarks
- Atomic Number
- Mass Number
- Building Atoms
- Atom Builder
- Gravitational Force
- Electromagnetic Force
- Strong Force
- Weak Force
- Isotopes
- Atomic Mass
- Ion
- Building Ions
Last added presentations
- Gravitation
- Mechanical, Electromagnetic, Electrical, Chemical and Thermal
- Newton's laws of motion
- Soil and Plant Nutrition
- The Effects of Radiation on Living Things
- Newton's Laws
- Radioactivity and Nuclear Reactions