The Reaction MechanismPage
1
1
Slide 1
Reaction Mechanism
The reaction mechanism is the series of elementary steps by which a chemical reaction occurs.
The sum of the elementary steps must give the overall balanced equation for the reaction
The mechanism must agree with the experimentally determined rate law
Slide 2
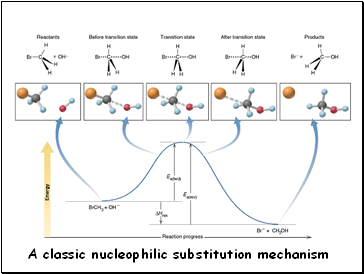
A classic nucleophilic substitution mechanism
Slide 3
Rate-Determining Step
In a multi-step reaction, the slowest step is the rate-determining step. It therefore determines the rate of the reaction.
The experimental rate law must agree with the rate-determining step
Slide 4
Identifying the Rate-Determining Step
For the reaction:
2H2(g) + 2NO(g) N2(g) + 2H2O(g)
The experimental rate law is:
R = k[NO]2[H2]
Which step in the reaction mechanism is the rate-determining (slowest) step?
Step #1 H2(g) + 2NO(g) N2O(g) + H2O(g)
Step #2 N2O(g) + H2(g) N2(g) + H2O(g)
Step #1 agrees with the experimental rate law
Slide 5
Identifying Intermediates
For the reaction:
2H2(g) + 2NO(g) N2(g) + 2H2O(g)
Which species in the reaction mechanism are intermediates (do not show up in the final, balanced equation?)
Step #1 H2(g) + 2NO(g) N2O(g) + H2O(g)
Step #2 N2O(g) + H2(g) N2(g) + H2O(g)
2H2(g) + 2NO(g) N2(g) + 2H2O(g)
N2O(g) is an intermediate
Slide 6
Collision Model
Key Idea: Molecules must collide to react.
However, only a small fraction of collisions produces a reaction. Why?
Slide 7
Collision Model
Collisions must have sufficient energy to produce the reaction (must equal or exceed the activation energy).
Colliding particles must be correctly oriented to one another in order to produce a reaction.
Slide 8
Factors Affecting Rate
Increasing temperature always increases the rate of a reaction.
Particles collide more frequently
Particles collide more energetically
Increasing surface area increases the rate of a reaction
Increasing Concentration USUALLY increases the rate of a reaction
Presence of Catalysts, which lower the activation energy by providing alternate pathways
Contents
- Reaction Mechanism
- Rate-Determining Step
- Identifying the Rate-Determining Step
- Identifying Intermediates
- Collision Model
- Factors Affecting Rate
Last added presentations
- Newton's Laws
- Heat-Energy on the Move
- Solar Thermal Energy
- Mechanics Lecture
- The Effects of Radiation on Living Things
- Radioactivity and Nuclear Reactions
- Gravitation