Transition elementsPage
1
1
Slide 1
The Transition Metals, the Lanthanides and the Antinides
Slide 2
The Metals in the Middle
Groups 3-12 are called the transition elements.
All of them are metals.
Transition Elements
3
Across any period from Group 3 through 12, the properties of the elements change less noticeably than they do across a period of representative elements.
Most transition elements are found combined with other elements in ores.
Slide 3
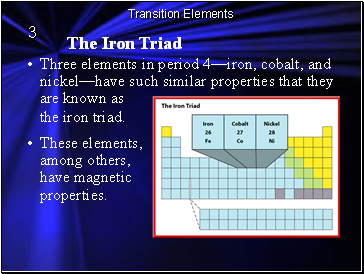
The Iron Triad
Transition Elements
3
These elements, among others, have magnetic properties.
Slide 4
The Iron Triad
Industrial magnets are made from an alloy of nickel, cobalt, and aluminum.
Transition Elements
3
Nickel is used in batteries along with cadmium.
Iron is a necessary part of hemoglobin, the substance that transports oxygen in the blood.
Iron also is mixed with other metals and with carbon to create a variety of steels with different properties.
Slide 5
Uses of Transition Elements
Most transition metals have higher melting points than the representative elements.
Transition Elements
3
Slide 6
Uses of Transition Elements
Transition Elements
3
Tungsten has the highest melting point of any metal (3,410°C) and will not melt when a current passes through it.
Slide 7
Uses of Transition Elements
Mercury, which has the lowest melting point of any metal (–39°C), is used in thermometers and in barometers.
Transition Elements
3
Mercury is the only metal that is a liquid at room temperatures.
Like many of the heavy metals, mercury is poisonous to living beings.
Slide 8
Uses of Transition Elements
Chromium’s name comes from the Greek word for color, chrome.
Transition Elements
3
Many other transition elements combine to form substances with brilliant colors.
Slide 9
Uses of Transition Elements
Ruthenium, rhodium, palladium, osmium, iridium, and platinum are sometimes called the platinum group because they have similar properties.
Transition Elements
3
They do not combine as easily with other elements.
As a result, they can be used as catalysts.
Contents
- The Metals in the Middle
- The Iron Triad
- Uses of Transition Elements
- Inner Transition Elements
- The Lanthanides
- The Actinides
- Dentistry and Dental Materials
Last added presentations
- Sensory and Motor Mechanisms
- Resource Acquisition and Transport in Vascular Plants
- Newton’s laws of motion
- Solar Thermal Energy
- Buoyancy
- Newton’s Laws of Motion
- Space Radiation