Bulk Properties of WaterPage
1
1
Slide 1
Properties of Water
The first image taken by humans of the whole Earth. Photographed by the crew of Apollo 8, the photo shows the Earth at a distance of about 30,000 km. Space has no respect for “North” and “South” as the southern most tip of South America is at the top of the photo.
Slide 2
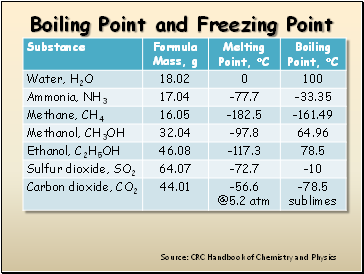
Boiling Point and Freezing Point
Source: CRC Handbook of Chemistry and Physics
Slide 3
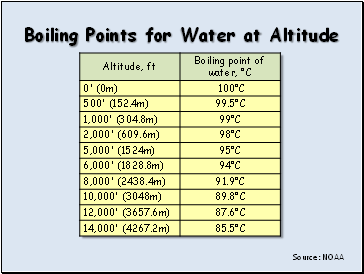
Boiling Points for Water at Altitude
Source: NOAA
Slide 4
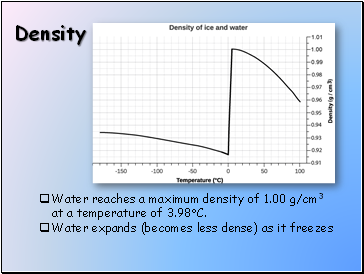
Density
Water reaches a maximum density of 1.00 g/cm3 at a temperature of 3.98C.
Water expands (becomes less dense) as it freezes
Slide 5
Rock Weathering
The expansion of freezing water exerts sufficient force to fracture rock, and is a significant cause of rock weathering.
Slide 6
Water’s Thermochemistry
Specific heat and Latent heat of phase change, as they apply to water.
Slide 7
Units for Measuring Heat
The Joule is the SI system unit for measuring heat:
The calorie is the heat required to raise the temperature of 1 gram of water by 1 Celsius degree
The British thermal unit (BTU or Btu) is the amount of energy needed to heat one pound of water by one degree Fahrenheit.
Slide 8
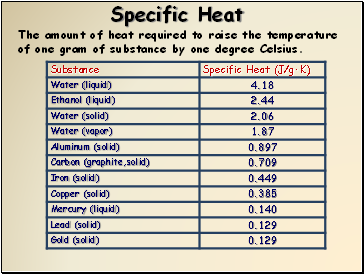
Specific Heat
The amount of heat required to raise the temperature of one gram of substance by one degree Celsius.
Slide 9
Specific Heat
The amount of heat required to raise the temperature of one gram of substance by one degree Celsius.
Slide 10
Specific Heat and Climate
How does water contribute to the moderation of climate in coastal communities?
Santa
Barbara
CA
Slide 11
Calculations Involving Specific Heat
cp = Specific Heat
Q = Heat lost or gained
T = Temperature change
OR
m = Mass
Slide 12
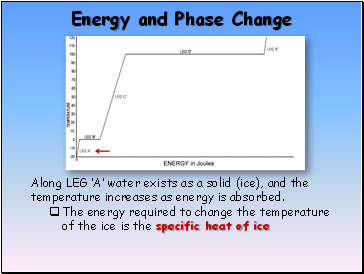
Energy and Phase Change
Along LEG ‘A’ water exists as a solid (ice), and the temperature increases as energy is absorbed.
Contents
- Properties of Water
- Boiling Point and Freezing Point
- Boiling Points for Water at Altitude
- Density
- Water’s Thermochemistry
- Units for Measuring Heat
- Specific Heat
- Specific Heat and Climate
- Calculations Involving Specific Heat
- Energy and Phase Change
- Heat of Fusion
- Heat of Vaporization
- Latent Heat – Sample Problem
Last added presentations
- Newton’s laws of motion
- Solar Thermal Energy
- Friction
- Newton’s Law of Gravity
- Mechanics Lecture
- Newton's laws of motion
- History of Modern Astronomy