Chemical EquilibriumPage
2
2
For the gas phase reaction:
3H2(g) + N2(g) 2NH3(g)
Slide 12
Heterogeneous Equilibria
The position of a heterogeneous equilibrium does not depend on the amounts of pure solids or liquids present
Write the equilibrium expression for the reaction:
PCl5(s) PCl3(l) + Cl2(g)
Pure
solid
Pure
liquid
Slide 13
The Reaction Quotient
For some time, t, when the system is not at equilibrium, the reaction quotient, Q takes the place of K, the equilibrium constant, in the law of mass action.
jA + kB lC + mD
Slide 14
Significance of the Reaction Quotient
If Q = K, the system is at equilibrium
If Q > K, the system shifts to the left, consuming products and forming reactants until equilibrium is achieved
If Q < K, the system shifts to the right, consuming reactants and forming products until equilibrium is achieved
Slide 15
Solving for Equilibrium Concentration
Consider this reaction at some temperature:
H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0
Assume you start with 8 molecules of H2O and 6 molecules of CO. How many molecules of H2O, CO, H2, and CO2 are present at equilibrium?
Here, we learn about “ICE” – the most important problem solving technique in the second semester. You will use it for the next 4 chapters!
Slide 16
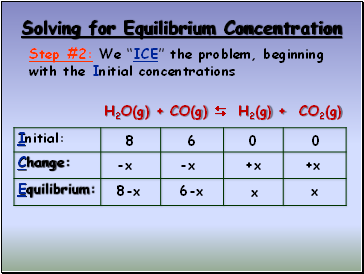
Solving for Equilibrium Concentration
H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0
Step #1: We write the law of mass action for the reaction:
Slide 17
Solving for Equilibrium Concentration
H2O(g) + CO(g) H2(g) + CO2(g)
Step #2: We “ICE” the problem, beginning with the Initial concentrations
8
6
0
0
-x
-x
+x
+x
8-x
6-x
x
x
Slide 18
Solving for Equilibrium Concentration
Step #3: We plug equilibrium concentrations into our equilibrium expression, and solve for x
H2O(g) + CO(g) H2(g) + CO2(g)
x = 4
Slide 19
Solving for Equilibrium Concentration
Step #4: Substitute x into our equilibrium concentrations to find the actual concentrations
H2O(g) + CO(g) H2(g) + CO2(g)
x = 4
1 2
Contents
- Chemical equilibrium
- Law of Mass Action
- Product Favored Equilibrium
- Reactant Favored Equilibrium
- Writing an Equilibrium Expression
- Conclusions about Equilibrium Expressions
- Equilibrium Expressions Involving Pressure
- Heterogeneous Equilibria
- The Reaction Quotient
- Significance of the Reaction Quotient
- Solving for Equilibrium Concentration
Last added presentations
- Heat-Energy on the Move
- Buoyancy
- Practical Applications of Solar Energy
- Friction
- Newton's laws of motion
- The Effects of Radiation on Living Things
- Space Radiation