Fission and FusionPage
3
3
Slide 25
Energy from Fission
mass difference, m = total mass before fission – total mass after fission
m = 3.91815 x 10-25 – 3.91550 x 10-25
m = 2.65 x 10-28 kg
This reduction in mass results in the release of energy.
Slide 26
Energy Released
The energy released can be calculated using the equation:
E = mc2
Where:
E = energy released (J)
m = mass difference (kg)
c = speed of light in a vacuum (3 x 108 ms-1)
Slide 27
Energy from Fission
E = mc2
Calculate the energy released from the following fission reaction:
m = 2.65 x 10-28 kg
c = 3 x 108 ms-1
E = E
E = 2.65 x 10-28 x (3 x 108)2
E = 2.385 x 10-11 J
Slide 28
Energy from Fission
The energy released from this fission reaction does not seem a lot.
This is because it is produced from the fission of a single nucleus.
Large amounts of energy are released when a large number of nuclei undergo fission reactions.
Slide 29
Energy from Fission
Each uranium-235 atom has a mass of 3.9014 x 10-25 kg.
The total number of atoms in 1 kg of uranium-235 can be found as follows:
No. of atoms in 1 kg of uranium-235 = 1/3.9014 x 10-25
No. of atoms in 1 kg of uranium-235 = 2.56 x 1024 atoms
Slide 30
Energy from Fission
If one uranium-235 atom undergoes a fission reaction and releases 2.385 x 10-11 J of energy, then the amount of energy released by 1 kg of uranium-235 can be calculated as follows:
total energy = energy per fission x number of atoms
total energy = 2.385 x 10-11 x 2.56 x 1024
total energy = 6.1056 x 1013 J
Slide 31
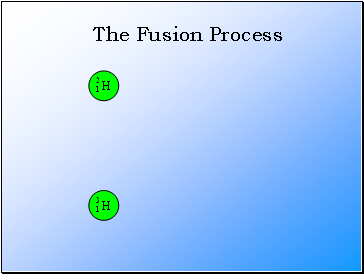
Nuclear Fusion
In nuclear fusion, two nuclei with low mass numbers combine to produce a single nucleus with a higher mass number.
Slide 32
The Fusion Process
Slide 33
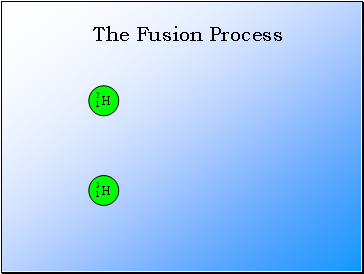
The Fusion Process
Slide 34
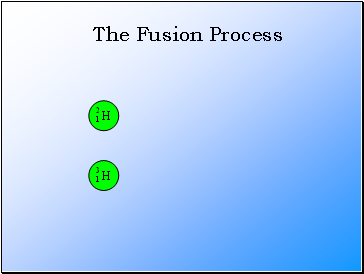
The Fusion Process
Slide 35
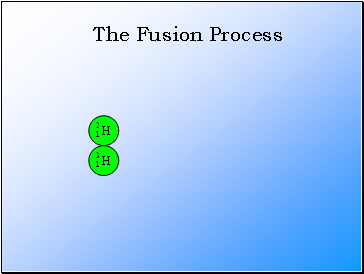
The Fusion Process
Slide 36
Contents
- Nuclear Reactions
- Fission
- Nuclear Fission
- Spontaneous Fission
- Induced Fission
- The Fission Process
- Energy from Fission
- Energy Released
- Nuclear Fusion
Last added presentations
- Space Radiation
- Newton’s third law of motion
- Radiation Safety and Operations
- Health Physics
- Madame Marie Curie
- Newton's laws of motion
- Heat-Energy on the Move