Elements compounds and mixturesPage
3
3
Lead has two charges listed, +2 and +4. This is a sample of lead (II) chloride (PbCl2). Two or more elements bonded in a whole-number ratio is a COMPOUND.
This compound is formed from the +4 version of lead. This is lead (IV) chloride (PbCl4). Notice how both samples of lead compounds have consistent composition throughout? Compounds are homogeneous!
منگل، 18 ذو القعد، 1436
14
Slide 15
Types of Compounds
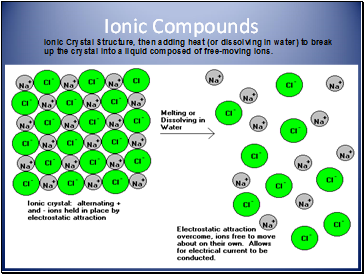
Ionic: made of metal and nonmetal ions. Form an ionic crystal lattice when in the solid phase. Ions separate when melted or dissolved in water, allowing electrical conduction. Examples: NaCl, K2O, CaBr2
Molecular: made of nonmetal atoms bonded to form a distinct particle called a molecule. Bonds do not break upon melting or dissolving, so molecular substances do not conduct electricity. EXCEPTION: Acids [H+A- (aq)] ionize in water to form H3O+ and A-, so they do conduct.
Network: made up of nonmetal atoms bonded in a seemingly endless matrix of covalent bonds with no distinguishable molecules. Very high m.p., don’t conduct.
منگل، 18 ذو القعد، 1436
15
Slide 16
Ionic Compounds
منگل، 18 ذو القعد، 1436
16
Slide 17
Molecular Compounds
منگل، 18 ذو القعد، 1436
17
Slide 18
Network Solids
Network solids are made of nonmetal atoms covalently bonded together to form large crystal lattices. No individual molecules can be distinguished. Examples include SiO2 (quartz). Corundum (Al2O3) also forms these, even though Al is considered a metal. Network solids are among the hardest materials known. They have extremely high melting points and do not conduct electricity.
منگل، 18 ذو القعد، 1436
18
Slide 19
Examples of some formula
19
منگل، 18 ذو القعد، 1436
Slide 20
Mixtures
Heterogeneous mixtures are not uniform throughout.
Homogeneous mixtures are uniform throughout.
Homogeneous mixtures are called solutions.
20
منگل، 18 ذو القعد، 1436
Contents
- Classification of Matter
- Pure Substances and Mixtures
- Characteristics of pure & impure substances
- Elements
- Classification of Elements as Metals & Non- Metals
- Elements & symbols
- Compounds
- Characteristics of compound
- Compounds
- Types of Compounds
- Ionic Compounds
- Molecular Compounds
- Network Solids
- Examples of some formula
- Mixtures
- Characteristics of mixture
- Difference between compounds & mixtures
- Physical vs. Chemical Properties
- Physical and Chemical Changes
- Testing the purity of a substance
Last added presentations
- Sensory and Motor Mechanisms
- History of Modern Astronomy
- Motion
- Solar Thermal Energy
- Magnetic field uses sound waves to ignite sun's ring of fire
- Understanding Heat Transfer, Conduction, Convection and Radiation
- Static and Kinetic Friction