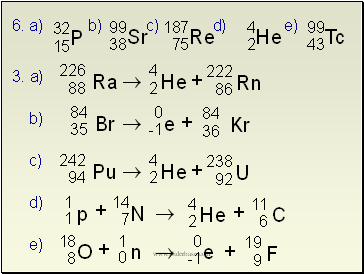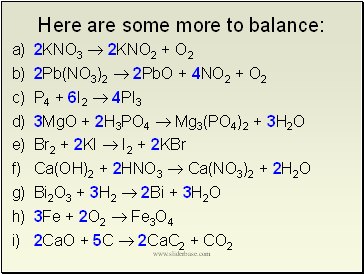Chemical and NuclearPage
2
2
Li + ZnCO3
Li + H2O
Al + O2
3
3
3
2
2
3
2
½
2
2
2
3
2
4
Slide 7
Discovery of Radioactivity
Radioactivity is the release of energy or particles when an atom disintegrates (demo)
Radioactivity was discovered when minerals were exposed to film through an opaque cover
The 3 types of radioactivity can be shown by passing emissions through an electrical field:
Slide 8
Types of Radioactivity
Types of radiation: 1) a, 2) b, 3)
Alpha () particles are symbolized as 42He
Beta () particles (essentially electrons) are 0–1e
Gamma (g) rays are symbolized as 00
You can determine the composition of each: a: mass of 4 u, charge of +2 (2 p+, 2 n0, 0 e–)
Other symbols: proton = 11p, neutron = 10n
There are different terms to describe the different types of nuclear reactions
“alpha decay” means an a particle is given off.
Other: beta decay, fusion (meaning to bring together), fission (meaning to break apart)
Slide 9
Nuclear equations
Q. Write the beta decay for C-14
In all cases, charge and mass must be balanced
Practice: pg. 222-3, Q6, Q3
C
14
6
e
0
-1
N
14
7
+
Po
209
84
He
4
2
Pb
205
82
+
Q. Write the alpha decay for 209Po
Q. Complete this fission reaction
Kr
94
36
Slide 10
6. a) b) c) d) e)
a)
b)
c)
d)
e)
Slide 11
a) 2KNO3 2KNO2 + O2
b) 2Pb(NO3)2 2PbO + 4NO2 + O2
c) P4 + 6I2 4PI3
d) 3MgO + 2H3PO4 Mg3(PO4)2 + 3H2O
e) Br2 + 2KI I2 + 2KBr
f) Ca(OH)2 + 2HNO3 Ca(NO3)2 + 2H2O
g) Bi2O3 + 3H2 2Bi + 3H2O
h) 3Fe + 2O2 Fe3O4
i) 2CaO + 5C 2CaC2 + CO2
Here are some more to balance:
Slide 12
Question 3 pg. 252
a) 2Li + 2H2O ® H2 + 2LiOH
b) P4 + 5O2 ® P4O10
c) 2C2H6 + 7O2 ® 4CO2 + 6H2O
d) CS2 + 3O2 ® CO2 + 2SO2
e) 2AsCl3 + 3H2S ® As2S3 + 6HCl
f) 3AgNO3 + FeCl3 ® 3AgCl + Fe(NO3)3
g) 2KClO3 ® 2KCl + 3O2
h) 2SO2 + O2 ® 2SO3
For more lessons, visit www.chalkbored.com
1 2
Contents
- How molecules are symbolized
- Balancing equations: MgO
- Balance equations by “inspection”
- Balance these skeleton equations:
- Returning to reaction types
- Discovery of Radioactivity
- Types of Radioactivity
- Nuclear equations
Last added presentations
- Gravitation
- Practical Applications of Solar Energy
- Solar Energy
- Static and Kinetic Friction
- Magnetic field uses sound waves to ignite sun's ring of fire
- Space Radiation
- Radioactivity and Nuclear Reactions