The Atom Atomic Number and Mass Number IsotopesPage
2
2
16 31 65
O P Zn
8 15 30
8 p+ 15 p+ 30 p+
8 n 16 n 35 n
8 e- 15 e- 30 e-
Slide 14
Isotopes
Atoms with the same number of protons, but different numbers of neutrons.
Atoms of the same element (same atomic number) with different mass numbers
Isotopes of chlorine
35Cl 37Cl
17 17
chlorine - 35 chlorine - 37
Slide 15
Learning Check
Naturally occurring carbon consists of three isotopes, 12C, 13C, and 14C. State the number of protons, neutrons, and electrons in each of these carbon atoms.
12C 13C 14C
6 6 6
#p _ _ _
#n _ _ _
#e _ _ _
Slide 16
Solution
12C 13C 14C
6 6 6
#p 6 6 6
#n 6 7 8
#e 6 6 6
Slide 17
Learning Check
An atom of zinc has a mass number of 65.
A. Number of protons in the zinc atom
1) 30 2) 35 3) 65
B. Number of neutrons in the zinc atom
1) 30 2) 35 3) 65
C. What is the mass number of a zinc isotope
with 37 neutrons?
1) 37 2) 65 3) 67
Slide 18
Solution
An atom of zinc has a mass number of 65.
A. Number of protons in the zinc atom
1) 30
B. Number of neutrons in the zinc atom
2) 35
C. What is the mass number of a zinc isotope
with 37 neutrons?
3) 67
Slide 19
Learning Check
Write the atomic symbols for atoms with the following:
A. 8 p+, 8 n, 8 e- _
B. 17p+, 20n, 17e- _
C. 47p+, 60 n, 47 e- _
Slide 20
Solution
16O
A. 8 p+, 8 n, 8 e- 8
B. 17p+, 20n, 17e- 37Cl
17
C. 47p+, 60 n, 47 e- 107Ag
47
Slide 21
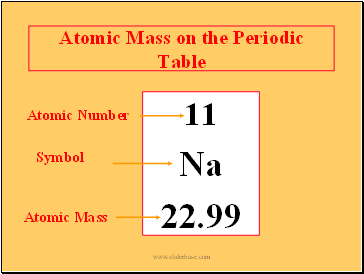
Atomic Mass on the Periodic Table
11
Na
22.99
Atomic Number
Symbol
Atomic Mass
Slide 22
Atomic Mass
Atomic mass is the weighted average mass of all the atomic masses of the isotopes of that atom.
Slide 23
Example of an Average Atomic Mass
Cl-35 is about 75.5 % and Cl-37 about 24.5% of natural chlorine.
35 x 75.5 = 26.4
100
35.5
37 x 24.5 = 9.07
100
1 2
Contents
- Atomic Theory
- Subatomic Particles
- Location of Subatomic Particles
- Atomic Number
- Atomic Number on the Periodic Table
- All atoms of an element have the same number of protons
- Learning Check
- Solution
- Mass Number
- Atomic Symbols
- Number of Electrons
- Subatomic Particles in Some Atoms
- Isotopes
- Atomic Mass on the Periodic Table
- Atomic Mass
Last added presentations
- Practical Applications of Solar Energy
- Mechanics Lecture
- Newton's laws of motion
- Newton’s Law of Gravity
- Newton’s third law of motion
- Static and Kinetic Friction
- Radiation