Valence ElectronsPage
1
1
Slide 1
Valence Electrons:
ELECTRONS AVAILABLE FOR BONDING
Slide 2
CA Standards
Students know how to use the periodic table to determine the number of electrons available for bonding.
Students know how to draw Lewis dot structures.
Slide 3
Definition
Valence electrons are electrons in the outmost shell (energy level). They are the electrons available for bonding.
Slide 4
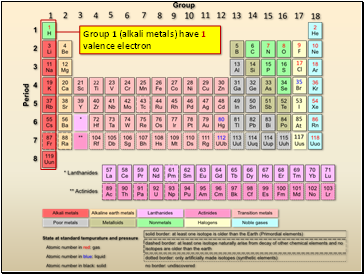
Group 1 (alkali metals) have 1 valence electron
Slide 5
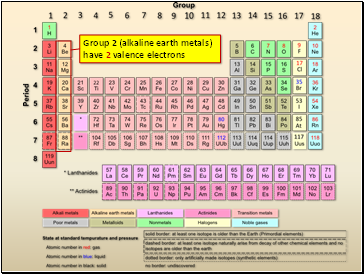
Group 2 (alkaline earth metals) have 2 valence electrons
Slide 6
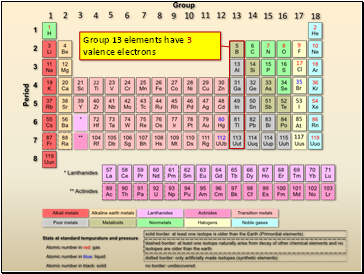
Group 13 elements have 3 valence electrons
Slide 7
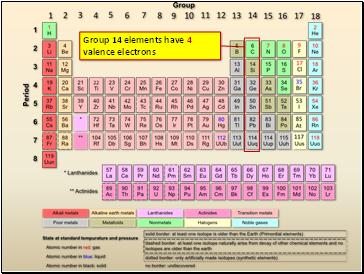
Group 14 elements have 4 valence electrons
Slide 8
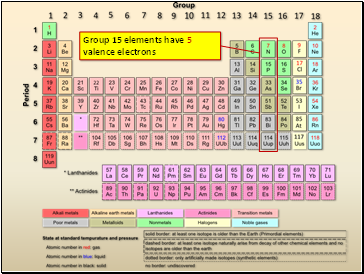
Group 15 elements have 5 valence electrons
Slide 9
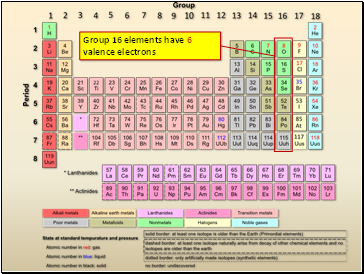
Group 16 elements have 6 valence electrons
Slide 10
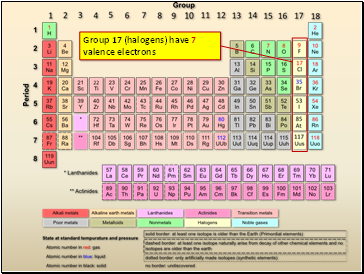
Group 17 (halogens) have 7 valence electrons
Slide 11
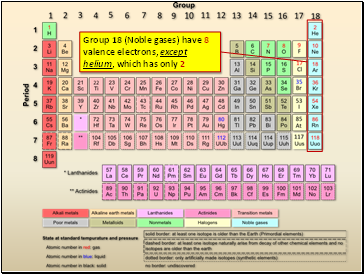
Group 18 (Noble gases) have 8 valence electrons, except helium, which has only 2
Slide 12
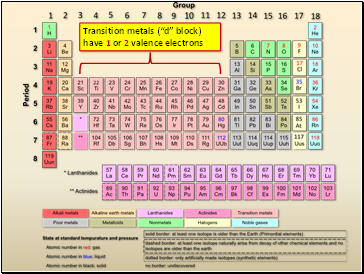
Transition metals (“d” block) have 1 or 2 valence electrons
Slide 13
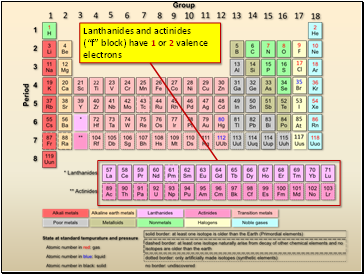
Lanthanides and actinides
(“f” block) have 1 or 2 valence electrons
Slide 14
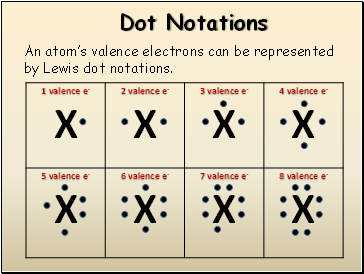
Dot Notations
An atom’s valence electrons can be represented by Lewis dot notations.
Slide 15
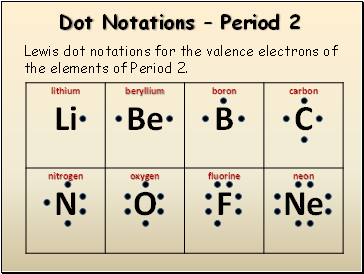
Dot Notations – Period 2
Lewis dot notations for the valence electrons of the elements of Period 2.
Contents
Last added presentations
- Soil and Plant Nutrition
- Direct heat utilization of geothermal energy
- Waves & Sound
- Sensory and Motor Mechanisms
- Newton's laws of motion
- Newton’s law of universal gravitation
- Newton’s Law of Gravity