EnzymesPage
1
1
Slide 1
Enzymes
Mr Potter
Slide 2
Lesson Objectives
Enzyme unit overview
What are they?
How they work
Activation energy
What controls their activity
Rates of reaction
Substrate/enzyme concentrations
Temperature, pH
Enzyme inhibitors
Practical to demonstrate “Catalase” activity in different tissue samples
Slide 3
Previous related topics covered?
Enzyme controlled reactions?
Proteins?
Lipase, protease, pectinase, amylase etc?
“Lock & Key” molecular structures?
Slide 4
By the end of the unit you should be able to:-
Explain enzymes as Globular Proteins which act as catalysts
Explain their catalytic action in terms of lowering activation energy
Describe examples of enzyme-catalysed reactions
Discuss factors affecting reaction rates and inhibition
Describe how to investigate these effects experimentally
Slide 5
Enzymes:-
Are defined as a BIOLOGICAL catalyst i.e. something that speeds up a reaction. Up to 1012 fold
Usually end in ‘…ase’.
Discovered in 1900 in yeasts. Some 40,000 in human cells
Control almost every metabolic reaction in living organisms
Are globular proteins coiled into a very precise 3-dimentional shape with hydrophilic side chains making them soluble
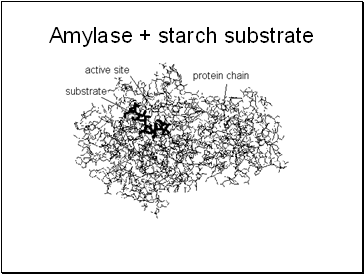
Possess an active site such as a cleft in the molecule onto which other substrate molecules can bind to form an enzyme-substrate complex
Once the substrate has been either synthesised or split, enzymes can be re-used.
Do not ‘create’ reactions
Widely used in industrial cleaning
Often require co-factors (co-enzymes) to function – metal ions, or vitamin derivatives
Slide 6
Slide 7
Amylase + starch substrate
Slide 8
How do enzymes work?
Reaction Mechanism
In any chemical reaction a substrate is converted into a product.
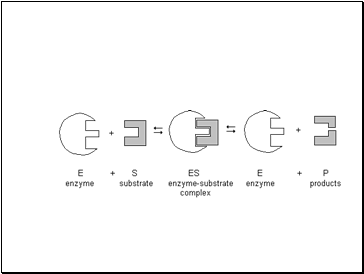
In an enzyme catalysed reaction the substrate first binds to the active site of the enzyme to form the enzyme-substrate complex
Slide 9
Slide 10
Molecule Geometry
Substrate molecule fits into the enzyme like a lock & key.
1 2
Contents
- Lesson Objectives
- Previous related topics covered?
- By the end of the unit you should be able to:-
- Amylase + starch substrate
- How do enzymes work?
- Molecule Geometry
- “Activation Energy”
Last added presentations
- Radiation
- Sensory and Motor Mechanisms
- Friction
- Newton's laws of motion
- Newton’s Law of Gravity
- Direct heat utilization of geothermal energy
- Motion