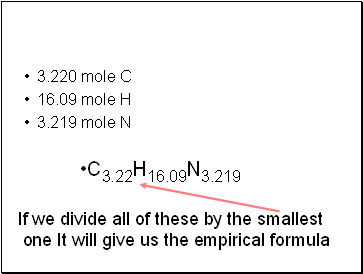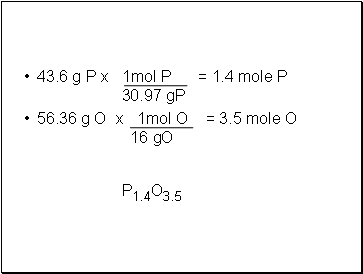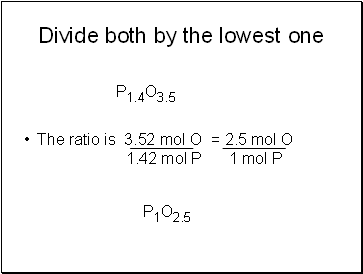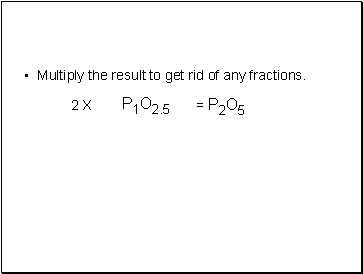Empirical FormulaPage
1
1
Slide 1
Empirical Formula
From percentage to formula
Slide 2
The Empirical Formula
The lowest whole number ratio of elements in a compound.
The molecular formula the actual ratio of elements in a compound
The two can be the same.
CH2 empirical formula
C2H4 molecular formula
C3H6 molecular formula
H2O both
Slide 3
Calculating Empirical
Just find the lowest whole number ratio
C6H12O6
CH4N
It is not just the ratio of atoms, it is also the ratio of moles of atoms
In 1 mole of CO2 there is 1 mole of carbon and 2 moles of oxygen
In one molecule of CO2 there is 1 atom of C and 2 atoms of O
Slide 4
Calculating Empirical
Pretend that you have a 100 gram sample of the compound.
That is, change the % to grams.
Convert the grams to mols for each element.
Write the number of mols as a subscript in a chemical formula.
Divide each number by the least number.
Multiply the result to get rid of any fractions.
Slide 5
Example
Calculate the empirical formula of a compound composed of 38.67 % C, 16.22 % H, and 45.11 %N.
Assume 100 g so
38.67 g C x 1mol C = 3.220 mole C 12.01 gC
16.22 g H x 1mol H = 16.09 mole H 1.01 gH
45.11 g N x 1mol N = 3.219 mole N 14.01 gN
Slide 6
3.220 mole C
16.09 mole H
3.219 mole N
C3.22H16.09N3.219
If we divide all of these by the smallest
one It will give us the empirical formula
Slide 7
Example
The ratio is 3.220 mol C = 1 mol C 3.219 molN 1 mol N
The ratio is 16.09 mol H = 5 mol H 3.219 molN 1 mol N
C1H5N1 is the empirical formula
A compound is 43.64 % P and 56.36 % O. What is the empirical formula?
Slide 8
43.6 g P x 1mol P = 1.4 mole P 30.97 gP
56.36 g O x 1mol O = 3.5 mole O 16 gO
P1.4O3.5
Slide 9
Divide both by the lowest one
The ratio is 3.52 mol O = 2.5 mol O 1.42 mol P 1 mol P
P1.4O3.5
P1O2.5
Slide 10
Multiply the result to get rid of any fractions.
P1O2.5
2 X
= P2O5
Slide 11
1 2
Contents
Last added presentations
- Magnetic field uses sound waves to ignite sun's ring of fire
- Ch 9 Nuclear Radiation
- Sensory and Motor Mechanisms
- Sound
- Solar Thermal Energy
- Newton’s Law of Gravity
- Waves & Sound