Acids and BasesPage
3
3
Slide 17

Solving Weak Acid Equilibrium Problems (continued)
Define change at equilibrium (as “x”).
Write equilibrium concentrations in terms of x.
Substitute equilibrium concentrations into equilibrium expression.
Solve for x the “easy way.”
Verify assumptions using 5% rule.
Calculate [H+] and pH.
Slide 18

Percent Dissociation (Ionization)
It is useful to specify the amount of weak acid that has dissociated in achieving equilibrium in an aqueous solution. The percent dissociation is defined as follows:
For a given weak acid, the percent dissociation increases as the acid becomes more dilute.
Slide 19
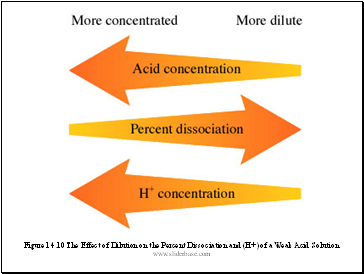
Figure 14.10 The Effect of Dilution on the Percent Dissociation and (H+) of a Weak Acid Solution
Slide 20

Bases
Arrhenius concept: A base is a substance that produces OH- ions in aqueous solution.
Bronsted-Lowry concept: A base is a proton acceptor.
“Strong” and “weak” are used in the same sense for bases as for acids.
strong = complete dissociation (hydroxide ion supplied to solution)
NaOH(s) Na+(aq) + OH(aq)
Slide 21

Bases (continued)
weak = very little dissociation (or reaction with water)
H3CNH2(aq) + H2O(l) H3CNH3+(aq) + OH(aq)
H3CNH2 molecule accepts a proton and thus functions as a base. Water is the acid in this reaction. Methyl amine contains no hydroxide ion, it still increases the concentration of hydroxide ion to yield a basic solution.
Slide 22

Polyprotic Acids
. . . can furnish more than one proton (H+) to the solution. A polyprotic acid always dissociates in a stepwise manner, one proton at a time.
For a typical weak polyprotic acid,
Ka1 > Ka2 > Ka3
Slide 23
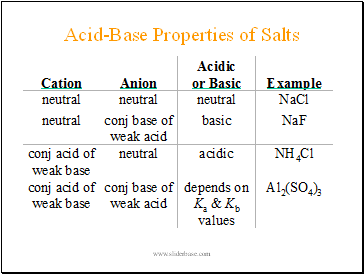
Acid-Base Properties of Salts
Slide 24

Structure and Acid-Base Properties
When a substance is dissolved in water, it produces an acidic solution if it can donate protons and produces a basic solution if it can accept protons.
Two factors for acidity in binary compounds:
Bond Polarity (high is good)
Bond Strength (low is good)
Slide 25
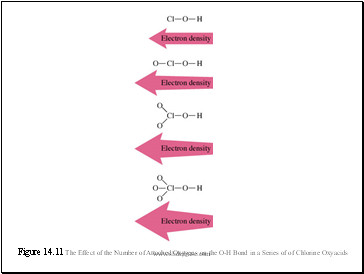
Figure 14.11 The Effect of the Number of Attached Oxygens on the O-H Bond in a Series of of Chlorine Oxyacids
Contents
- Acids and Bases
- Conjugate Acid/Base Pairs
- Acid Dissociation Constant (Ka)
- Acid Strength
- Water as an Acid and a Base
- The pH Scale
- Calculating the pH of Strong Acid Solutions
- Solving Weak Acid Equilibrium Problems
- Percent Dissociation (Ionization)
- Bases
- Polyprotic Acids
- Acid-Base Properties of Salts
- Structure and Acid-Base Properties
- Oxides
- Lewis Acids and Bases
Last added presentations
- Madame Marie Curie
- Sound
- Newton’s laws of motion
- Radiation
- Static and Kinetic Friction
- Simulation at NASA for the Space Radiation Effort
- Heat-Energy on the Move
