TLC and GLCPage
1
1
Slide 1
Chromatography
Is a technique used to separate and identify the components of a mixture.
Works by allowing the molecules present in the mixture to distribute themselves between a stationary and a mobile medium.
Molecules that spend most of their time in the mobile phase are carried along faster.
Slide 2
Gas Liquid Chromatography
Here the mobile phase is an unreactive gas ( eg Nitrogen) flowing through a tube.
And the stationary phase is an involatile liquid held on particles of a solid support.
Slide 3
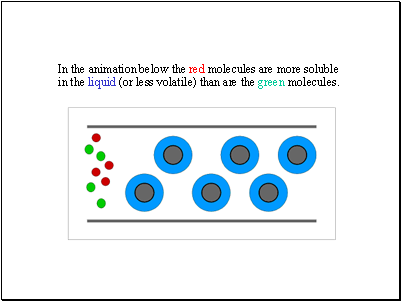
In the animation below the red molecules are more soluble in the liquid (or less volatile) than are the green molecules.
Slide 4
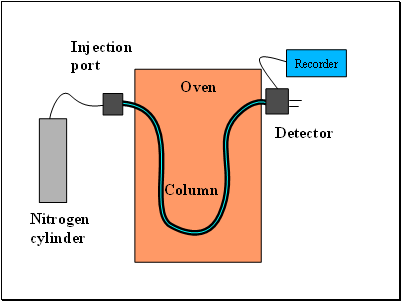
In practice the Column is contained in a thermostatic oven. (Why ?)
About 1μL of liquid is injected into one end of the column.
As each component reaches the other end it is detected and registered on a chart recorder.
The Retention Time is characteristic of a particular substance. (for the same column, temperature, gas flow etc.)
The area under each peak indicates the relative quantities.
Slide 5
Oven
Detector
Injection
port
Nitrogen
cylinder
Column
Recorder
Slide 6
Slide 7
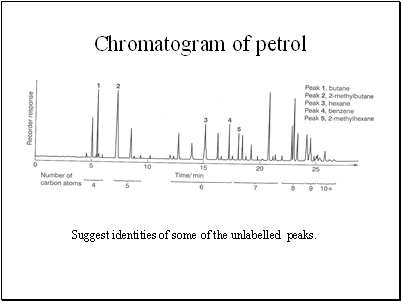
Chromatogram of petrol
Suggest identities of some of the unlabelled peaks.
Slide 8
Thin Layer Chromatography
Here the mobile phase is a liquid
Flowing past a thin layer of powder on a solid support.
Substances that are less attracted to the solid or are more soluble in the liquid move faster.
And so move further up the plate by the time that the process has been stopped by taking the plate out of the liqiud. - larger Rf
Slide 9
Slide 10
Rf = distance moved by substance
distance moved by solvent front
For substances that are very soluble in the liquid Rf will be close to
For substances that are rather insoluble in the liquid Rf will be close to
1
0
Contents
Last added presentations
- Thermal Energy
- Sensory and Motor Mechanisms
- Heat-Energy on the Move
- Sound
- Radiation Safety and Operations
- Ch 9 Nuclear Radiation
- Mechanics Lecture