Atomic Structure, History of the AtomPage
1
1
Slide 1
Atomic Structure
S.MORRIS 2006
More free powerpoints at www.worldofteaching.com
This Powerpoint is hosted on www.worldofteaching.com
Please visit for 100’s more free powerpoints
Slide 2
History of the atom
460 BC
Democritus develops the idea of atoms
he pounded up materials in his pestle and mortar until he had reduced them to smaller and smaller particles which he called
ATOMA
(greek for indivisible)
Slide 3
HISTORY OF THE ATOM
1808
John Dalton
suggested that all matter was made up of tiny spheres that were able to bounce around with perfect elasticity and called them
ATOMS
Slide 4
HISTORY OF THE ATOM
1898
Joseph John Thompson
found that atoms could sometimes eject a far smaller negative particle which he called an
ELECTRON
Slide 5
HISTORY OF THE ATOM
Thompson develops the idea that an atom was made up of electrons scattered unevenly within an elastic sphere surrounded by a soup of positive charge to balance the electron's charge
1904
like plums surrounded by pudding.
PLUM PUDDING
MODEL
Slide 6
HISTORY OF THE ATOM
1910
Ernest Rutherford
oversaw Geiger and Marsden carrying out his famous experiment.
they fired Helium nuclei at a piece of gold foil which was only a few atoms thick.
they found that although most of them passed through. About 1 in 10,000 hit
Slide 7
HISTORY OF THE ATOM
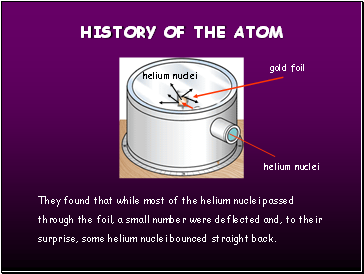
gold foil
helium nuclei
They found that while most of the helium nuclei passed through the foil, a small number were deflected and, to their surprise, some helium nuclei bounced straight back.
helium nuclei
Slide 8
HISTORY OF THE ATOM
Rutherford’s new evidence allowed him to propose a more detailed model with a central nucleus.
He suggested that the positive charge was all in a central nucleus. With this holding the electrons in place by electrical attraction
However, this was not the end of the story.
Slide 9
HISTORY OF THE ATOM
1913
Niels Bohr
studied under Rutherford at the Victoria University in Manchester.
Bohr refined Rutherford's idea by adding that the electrons were in orbits. Rather like planets orbiting the sun. With each orbit only able to contain a set number of electrons.
Contents
- Atomic Structure
- History of the atom
- Bohr’s Atom
- Helium atom
- Atomic structure
- Electronic configuration
- Dot & Cross diagrams
- Summary
Last added presentations
- Sound
- Direct heat utilization of geothermal energy
- Sound
- Space Radiation
- History of Modern Astronomy
- Solar Energy
- Newton's Laws