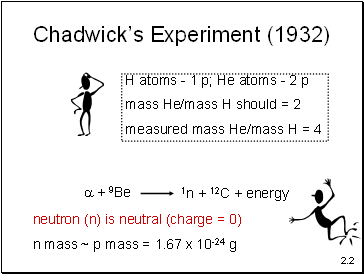Atoms, molecules and ionsPage
1
1
Slide 1
Atoms, Molecules and Ions
Chapter 2
Slide 2
Dalton’s Atomic Theory (1808)
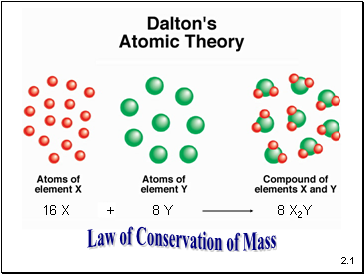
Elements are composed of extremely small particles called atoms. All atoms of a given element are identical. The atoms of one element are different from the atoms of all other elements.
Compounds are composed of atoms of more than one element. The relative number of atoms of each element in a given compound is always the same.
Chemical reactions only involve the rearrangement of atoms. Atoms are not created or destroyed in chemical reactions.
2.1
Slide 3
2
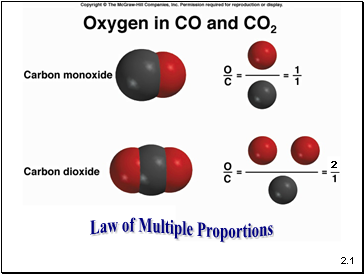
Law of Multiple Proportions
2.1
Slide 4
8 X2Y
Law of Conservation of Mass
2.1
Slide 5
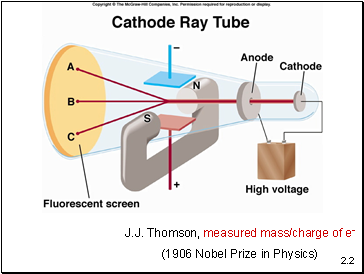
J.J. Thomson, measured mass/charge of e-
(1906 Nobel Prize in Physics)
2.2
Slide 6
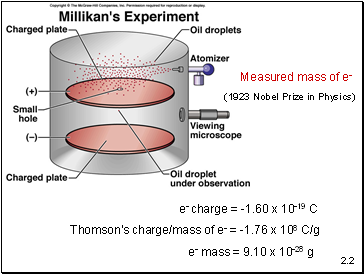
e- charge = -1.60 x 10-19 C
Thomson’s charge/mass of e- = -1.76 x 108 C/g
e- mass = 9.10 x 10-28 g
Measured mass of e-
(1923 Nobel Prize in Physics)
2.2
Slide 7
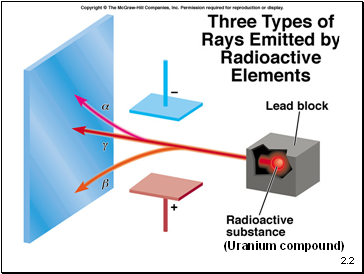
(Uranium compound)
2.2
Slide 8
2.2
Slide 9
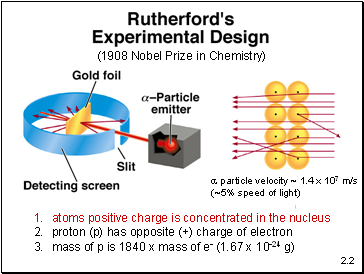
atoms positive charge is concentrated in the nucleus
proton (p) has opposite (+) charge of electron
mass of p is 1840 x mass of e- (1.67 x 10-24 g)
particle velocity ~ 1.4 x 107 m/s
(~5% speed of light)
(1908 Nobel Prize in Chemistry)
2.2
Slide 10
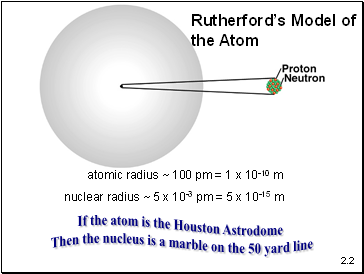
atomic radius ~ 100 pm = 1 x 10-10 m
nuclear radius ~ 5 x 10-3 pm = 5 x 10-15 m
Rutherford’s Model of
the Atom
If the atom is the Houston Astrodome
Then the nucleus is a marble on the 50 yard line
2.2
Slide 11
Chadwick’s Experiment (1932)
H atoms - 1 p; He atoms - 2 p
mass He/mass H should = 2
measured mass He/mass H = 4
neutron (n) is neutral (charge = 0)
n mass ~ p mass = 1.67 x 10-24 g
2.2
Slide 12
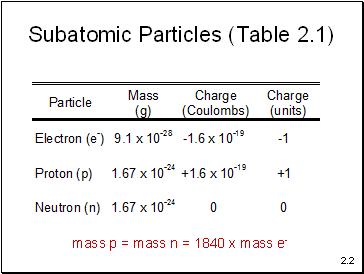
Subatomic Particles (Table 2.1)
mass p = mass n = 1840 x mass e-
2.2
Slide 13
Contents
- Atoms, Molecules and Ions
- Dalton’s Atomic Theory (1808)
- Chadwick’s Experiment (1932)
- Formula of Ionic Compounds
- Chemical Nomenclature
Last added presentations
- Radioactivity and Nuclear Reactions
- Motion
- Soil and Plant Nutrition
- Newton’s Laws of Motion
- Health Physics
- Direct heat utilization of geothermal energy
- Understanding Heat Transfer, Conduction, Convection and Radiation