Colligative Properties of SolutionsPage
1
1
Slide 1
Colligative Properties of Solutions
Jacobus Henricus van 't Hoff
(1852-1911)
Slide 2
Colligative Properties
Colligative properties are those that depend on the concentration of particles in a solution, not upon the identity of those particles.
Boiling Point Elevation
Freezing Point Depression
Osmotic Pressure
Slide 3
Freezing Point Depression
Each mole of solute particles lowers the freezing point of 1 kilogram of water by 1.86 degrees Celsius.
Kf = 1.86 C kilogram/mol
m = molality of the solution
i = van’t Hoff factor
Slide 4
Boiling Point Elevation
Each mole of solute particles raises the boiling point of 1 kilogram of water by 0.51 degrees Celsius.
Kb = 0.51 C kilogram/mol
m = molality of the solution
i = van’t Hoff factor
Slide 5
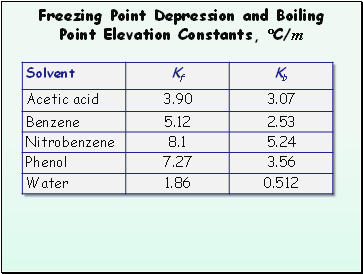
Freezing Point Depression and Boiling Point Elevation Constants, C/m
Slide 6
The van’t Hoff Factor, i
Electrolytes may have two, three or more times the effect on boiling point, freezing point, and osmotic pressure, depending on its dissociation.
Slide 7
Dissociation Equations and the Determination of i
NaCl(s)
AgNO3(s)
MgCl2(s)
Na2SO4(s)
AlCl3(s)
Na+(aq) + Cl-(aq)
Ag+(aq) + NO3-(aq)
Mg2+(aq) + 2 Cl-(aq)
2 Na+(aq) + SO42-(aq)
Al3+(aq) + 3 Cl-(aq)
i = 2
i = 2
i = 3
i = 3
i = 4
Slide 8
Ideal vs. Real van’t Hoff Factor
The ideal van’t Hoff Factor is only achieved in VERY DILUTE solution.
Slide 9
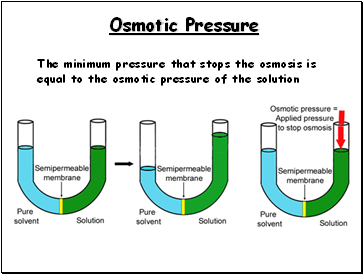
Osmotic Pressure
The minimum pressure that stops the osmosis is equal to the osmotic pressure of the solution
Slide 10
Osmotic Pressure Calculations
= Osmotic pressure
M = Molarity of the solution
R = Gas Constant = 0.08206 Latm/molK
i = van’t Hoff Factor
Contents
- Colligative Properties
- Freezing Point Depression
- Boiling Point Elevation
- The van’t Hoff Factor, i
- Dissociation Equations and the Determination of i
- Ideal vs. Real van’t Hoff Factor
- Osmotic Pressure
Last added presentations
- Simulation at NASA for the Space Radiation Effort
- Thermal Energy
- Madame Marie Curie
- Radioactivity and Nuclear Reactions
- Direct heat utilization of geothermal energy
- Newton’s Laws of Motion
- Mechanics Lecture