The Nature of Light and the Laws of Geometric OpticsPage
2
2
Some experiments could not be explained by the wave model of light.
The photoelectric effect was a major phenomenon not explained by waves.
When light strikes a metal surface, electrons are sometimes ejected from the surface.
The kinetic energy of the ejected electron is independent of the frequency of the light.
Einstein (in 1905) proposed an explanation of the photoelectric effect that used the idea of quantization.
The quantization model assumes that the energy of a light wave is present in particles called photons.
E = hƒ
h is Planck’s Constant and = 6.63 x 10-34 J.s
Section 35.1
Slide 8
Dual Nature of Light
In view of these developments, light must be regarded as having a dual nature.
Light exhibits the characteristics of a wave in some situations and the characteristics of a particle in other situations.
This chapter investigates the wave nature of light.
Section 35.1
Slide 9
Measurements of the Speed of Light
Since light travels at a very high speed, early attempts to measure its speed were unsuccessful.
Remember c = 3.00 x 108 m/s
Galileo tried by using two observers separated by about 10 km.
The reaction time of the observers was more than the transit time of the light.
Section 35.2
Slide 10
Measurement of the Speed of Light – Roemer’s Method
In 1675 Ole Roemer used astronomical observations to estimate the speed of light.
He used the period of revolution of Io, a moon of Jupiter, as Jupiter revolved around the sun.
The angle through which Jupiter moves during a 90° movement of the Earth was calculated.
Section 35.2
Slide 11
Roemer’s Method, cont.
The periods of revolution were longer when the Earth was receding from Jupiter.
Shorter when the Earth was approaching
Using Roemer’s data, Huygens estimated the lower limit of the speed of light to be 2.3 x 108 m/s.
This was important because it demonstrated that light has a finite speed as well as giving an estimate of that speed.
Section 35.2
Slide 12
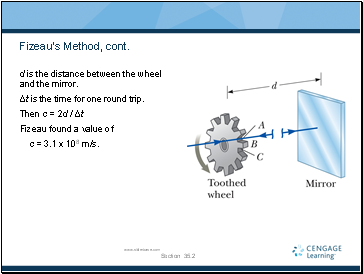
Measurements of the Speed of Light – Fizeau’s Method
This was the first successful method for measuring the speed of light by means of a purely terrestrial technique.
It was developed in 1849 by Armand Fizeau.
He used a rotating toothed wheel.
The distance between the wheel (considered to be the source) and a mirror was known.
Section 35.2
Slide 13
Fizeau’s Method, cont.
Contents
- Introduction to Light
- Light and Optics
- The Nature of Light
- Nature of Light – Alternative View
- Christian Huygens
- Confirmation of Wave Nature
- Particle Nature
- Dual Nature of Light
- Measurements of the Speed of Light
- Measurement of the Speed of Light – Roemer’s Method
- Roemer’s Method, cont.
- Measurements of the Speed of Light – Fizeau’s Method
- Fizeau’s Method, cont.
- The Ray Approximation in Ray Optics
- Ray Approximation
- Ray Approximation, cont.
- Reflection of Light
- Specular Reflection
- Diffuse Reflection
- Law of Reflection
- Law of Reflection, cont.
- Multiple Reflections
- Retroreflection
- Refraction of Light
- Refraction, cont.
- Refraction of Light, final
- Following the Reflected and Refracted Rays
- Refraction Details, 1
- Refraction Details, 2
- Light in a Medium
- The Index of Refraction
- Index of Refraction, cont.
- Frequency Between Media
- Index of Refraction Extended
- More About Index of Refraction
- Snell’s Law of Refraction
- Prism
- Huygens’s Principle
- Huygens’s Construction for a Plane Wave
- Huygens’s Construction for a Spherical Wave
- Huygens’s Principle and the Law of Reflection
- Huygens’s Principle and the Law of Reflection, cont.
- Huygens’s Principle and the Law of Refraction
- Huygens’s Principle and the Law of Refraction, cont.
- Huygens’s Principle and the Law of Refraction, final
- Dispersion
- Variation of Index of Refraction with Wavelength
- Refraction in a Prism
- The Rainbow
- The Rainbow, cont.
- Observing the Rainbow
- Double Rainbow
- Total Internal Reflection
- Possible Beam Directions
- Critical Angle
- Critical Angle, cont.
- Fiber Optics
- Construction of an Optical Fiber
- Fiber Optics, cont.
Last added presentations
- Soil and Plant Nutrition
- Radiation Safety and Operations
- Newton’s laws of motion
- Simulation at NASA for the Space Radiation Effort
- Solar Thermal Energy
- The Effects of Radiation on Living Things
- Geophysical Concepts, Applications and Limitations