Spontaneity, Entropy, and Free EnergyPage
1
1
Slide 1
Spontaneity, Entropy and Free Energy
Slide 2
Spontaneous Processes and Entropy
First Law
“Energy can neither be created nor destroyed"
The energy of the universe is constant
Spontaneous Processes
Processes that occur without outside intervention
Spontaneous processes may be fast or slow
Many forms of combustion are fast
Conversion of diamond to graphite is slow
Slide 3
Entropy (S)
A measure of the randomness or disorder
The driving force for a spontaneous process is an increase in the entropy of the universe
Entropy is a thermodynamic function describing the number of arrangements that are available to a system
Nature proceeds toward the states that have the highest probabilities of existing
Slide 4
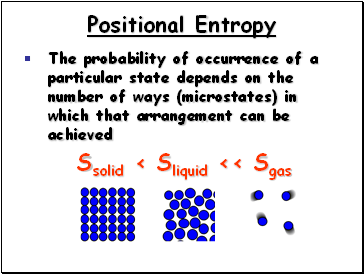
Positional Entropy
The probability of occurrence of a particular state depends on the number of ways (microstates) in which that arrangement can be achieved
Ssolid < Sliquid << Sgas
Slide 5
Second Law of Thermodynamics
"In any spontaneous process there is always an increase in the entropy of the universe"
"The entropy of the universe is increasing"
For a given change to be spontaneous, Suniverse must be positive
Suniv = Ssys + Ssurr
Slide 6
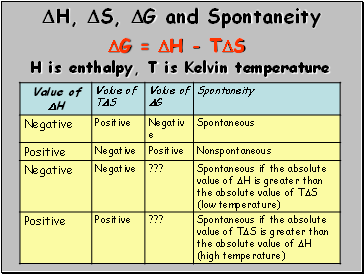
H, S, G and Spontaneity
G = H - TS
H is enthalpy, T is Kelvin temperature
Slide 7
Calculating Entropy Change in a Reaction
Entropy is an extensive property (a function of the number of moles)
Generally, the more complex the molecule, the higher the standard entropy value
Slide 8
Standard Free Energy Change
G0 is the change in free energy that will occur if the reactants in their standard states are converted to the products in their standard states
G0 cannot be measured directly
The more negative the value for G0, the farther to the right the reaction will proceed in order to achieve equilibrium
Equilibrium is the lowest possible free energy position for a reaction
Slide 9
For reactions at constant temperature:
1 2
Contents
- Spontaneous Processes and Entropy
- Entropy (S)
- Positional Entropy
- Second Law of Thermodynamics
- Calculating Entropy Change in a Reaction
- Standard Free Energy Change
- The Dependence of Free Energy on Pressure
- Free Energy and Equilibrium
- Temperature Dependence of K
- Free Energy and Work
Last added presentations
- Solar Energy
- Static and Kinetic Friction
- Radiation Safety and Operations
- Sensory and Motor Mechanisms
- Radioactivity and Nuclear Reactions
- Newton’s third law of motion
- Practical Applications of Solar Energy