Earth MaterialsPage
6
6
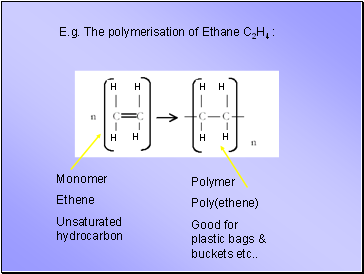
E.g. The polymerisation of Ethane C2H4 :
Monomer
Ethene
Unsaturated hydrocarbon
Polymer
Poly(ethene)
Good for plastic bags & buckets etc
Slide 44
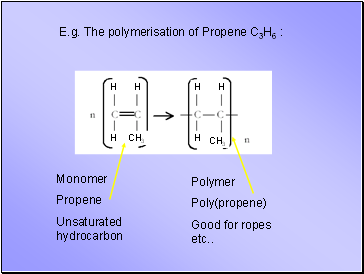
E.g. The polymerisation of Propene C3H6 :
Monomer
Propene
Unsaturated hydrocarbon
Polymer
Poly(propene)
Good for ropes etc
Slide 45
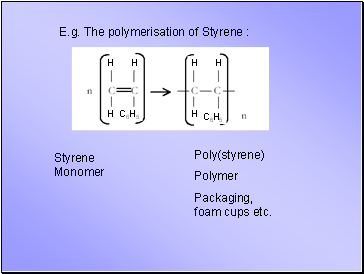
E.g. The polymerisation of Styrene :
Styrene Monomer
Poly(styrene)
Polymer
Packaging, foam cups etc.
Slide 46
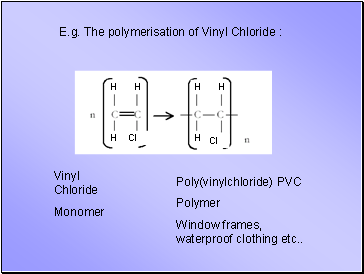
E.g. The polymerisation of Vinyl Chloride :
Vinyl Chloride
Monomer
Poly(vinylchloride) PVC
Polymer
Window frames, waterproof clothing etc
Slide 47
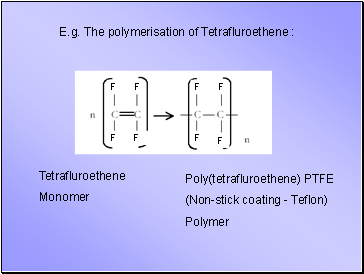
E.g. The polymerisation of Tetrafluroethene :
Poly(tetrafluroethene) PTFE
(Non-stick coating - Teflon)
Polymer
Tetrafluroethene
Monomer
Slide 48
Some uses of Plastics
Slide 49
Problems with Plastics
The co-valent bonds holding the long molecules together are very strong. Few bacteria can make them rot down. They tend not to be Biodegradable.(What does this word mean?)
This makes disposing of plastics difficult:
Burning them produces oxides of Carbon, Hydrogen and sometimes Sulphur and other toxic compounds:
Carbon Dioxide, Sulphur Dioxide, Water vapour
They fill up land-fill sites.
(Why should these factors be a problem?)
Slide 50
Why not recycle plastics?
Recycling plastics is complicated by several issues:
·Not all plastics lend themselves to recycling
·Different types of plastic have different properties, and so separation is very important.
·The economics of recycling are not always favourable, as the market is prone to fluctuate. For example, when oil prices fall the production of ‘new’ plastics become cheaper, so providing its recycled counterpart with greater competition.
·Unlike some countries a lack of subsidy in the UK makes a recycling industry more difficult to sustain, so nationally our recycling performance tends to lag behind many of our European neighbours.
However, increasing pressures on landfill sites, and our need to conserve natural resources makes recycling of plastics an important step forwards.
Slide 51
Why not burn them then?
Contents
- Earth Materials. Module 06
- Limestone
- Reminder:
- Cement
- Glass
- The Oil is often found in porous rock
- Reminder!
- More about Catalytic Cracking
- A Test for Unsaturated Hydrocarbons
- Why bother about Alkenes?
- Addition Polymerisation
- Ethene to Poly(ethene)
- Representing Polymerisation
- Some uses of Plastics
- Problems with Plastics
- Why not recycle plastics?
- Why not burn them then?
- The need for a balanced solution
Last added presentations
- History of Modern Astronomy
- Newton’s laws of motion
- Sound
- Sensory and Motor Mechanisms
- Newton's laws of motion
- Mechanical, Electromagnetic, Electrical, Chemical and Thermal
- Understanding Heat Transfer, Conduction, Convection and Radiation