Element ClassesPage
1
1
Slide 1
Element classes
Slide 2
CA Standards
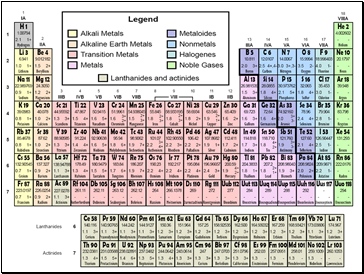
Students know how to use the periodic table to identify alkali metals, alkaline earth metals, transition metals, metals, semimetals (metalloids), nonmetals, halogens and noble gases.
Slide 3
Slide 4
Alkali Metals
All alkali metals have 1 valence electron
Alkali metals are NEVER found pure in nature; they are too reactive
Reactivity of these elements increases down the group
Potassium, K reacts with water and must be stored in kerosene
Slide 5
Alkaline Earth Metals
All alkaline earth metals have 2 valence electrons
Alkaline earth metals are less reactive than alkali metals
Alkaline earth metals are not found pure in nature; they are too reactive
The word “alkaline” means “basic”
common bases include salts of the metals
Ca(OH)2
Mg(OH)2
Slide 6
Properties of Metals
Metals are good conductors of heat and electricity
Metals are malleable
Metals are ductile
Metals have high tensile strength
Metals have luster
Slide 7
Transition Metals
Copper, Cu, is a relatively soft metal, and a very good electrical conductor.
Mercury, Hg, is the only metal that exists as a liquid at room temperature
Slide 8
Properties of Metalloids
They have properties of both metals and nonmetals.
Metalloids are more brittle than metals, less brittle than most nonmetallic solids
Metalloids are semiconductors of electricity
Some metalloids possess metallic luster
Slide 9
Silicon, Si – A Metalloid
Silicon has metallic luster
Silicon is brittle like a nonmetal
Silicon is a semiconductor of electricity
Other metalloids include:
Boron, B
Germanium, Ge
Arsenic, As
Antimony, Sb
Tellurium, Te
Slide 10
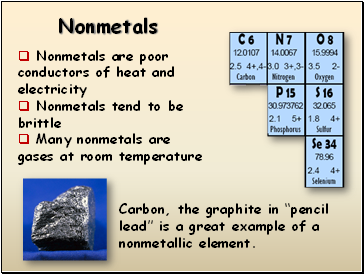
Nonmetals
Nonmetals are poor conductors of heat and electricity
Nonmetals tend to be brittle
Many nonmetals are gases at room temperature
Carbon, the graphite in “pencil lead” is a great example of a nonmetallic element.
Slide 11
1 2
Contents
- Element classes
- Alkali Metals
- Alkaline Earth Metals
- Properties of Metals
- Transition Metals
- Properties of Metalloids
- Silicon, Si – A Metalloid
- Nonmetals
- Examples of Nonmetals
- Halogens
- Noble Gases
Last added presentations
- Newton's laws of motion
- Ch 9 Nuclear Radiation
- Resource Acquisition and Transport in Vascular Plants
- Magnetic field uses sound waves to ignite sun's ring of fire
- Health Physics
- Madame Marie Curie
- Buoyancy