Atoms and the Periodic tablePage
9
9
Slide 82
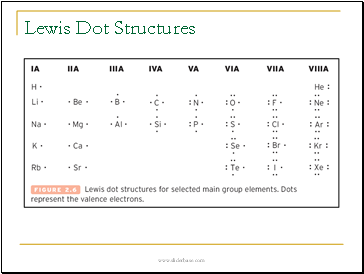
Writing Electron-Dot Symbols
Electron-dot symbols for
•groups 1A(1) to 4A(14) use single dots.
· ·
Na · · Mg · · Al · · C ·
·
•groups 5A(15) to 7A(17) use pairs and single dots.
· · · ·
· P · : O ·
· ·
Slide 83
Groups and Electron-Dot Symbols
In a group, all the electron-dot symbols have the
same number of valence electrons (dots).
Example: Atoms of elements in Group 2A(2) each have 2 valence electrons.
· Be ·
· Mg ·
· Ca ·
· Sr ·
· Ba ·
Slide 84
Lewis Dot Structures
Slide 85
Learning Check
A. X is the electron-dot symbol for
1) Na 2) K 3) Al
l
B. l X l
l is the electron-dot symbol of
1) B 2) N 3) P
Slide 86
Solution
l
A. X is the electron-dot symbol for
1) Na 2) K
B. l X l
l is the electron-dot symbol of
2) N 3) P
Slide 87
Chapter Outline
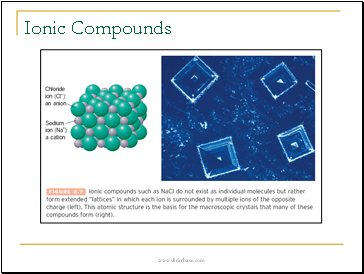
Ionic Bonds
Ionic compounds result from the loss of electrons by one atom (usually a metal) and the gain of electrons by another atom (usually a nonmetal).
Ionic bonds arise from the attraction between particles with opposite charges (electrostatic forces); e.g., Na+ Cl-.
Slide 88
Run the following web animations/movies.
3.7: Ion Formation
Slide 89
Ionic Compounds
Slide 90
Atomic Size
Atomic size is described using the atomic radius; the distance from the nucleus to the valence electrons.
Slide 91
Atomic Radius Within A Group
Atomic radius increases going down each group of representative elements.
Slide 92
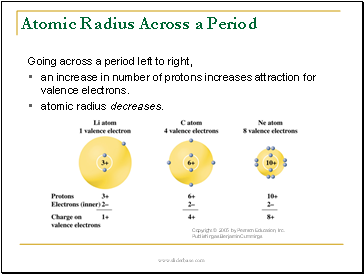
Atomic Radius Across a Period
Going across a period left to right,
•an increase in number of protons increases attraction for valence electrons.
•atomic radius decreases.
Slide 93
Contents
- Atoms and the periodic table
- Conservation of Matter
- Atomic Line Spectra
- Atomic Subshell Energies
- Exploration of the Periodic Table/ Periodic Reactivity Trends
Last added presentations
- Resource Acquisition and Transport in Vascular Plants
- Radiation
- Buoyancy
- Ch 9 Nuclear Radiation
- Sound
- Heat-Energy on the Move
- Newton’s Laws of Motion