Properties of SolutionsPage
1
1
Slide 1
Properties of Solutions
Slide 2
CA Standards
Slide 3
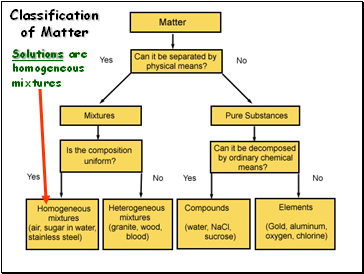
Classification of Matter
Solutions are homogeneous mixtures
Slide 4
Solute
A solute is the dissolved substance in a solution.
A solvent is the dissolving medium in a solution.
Solvent
Salt in salt water
Sugar in soda drinks
Carbon dioxide in soda drinks
Water in salt water
Water in soda
Slide 5
“Like Dissolves Like”
Polar and ionic solutes dissolve best in polar solvents
Nonpolar solutes dissolve best in nonpolar solvents
Slide 6
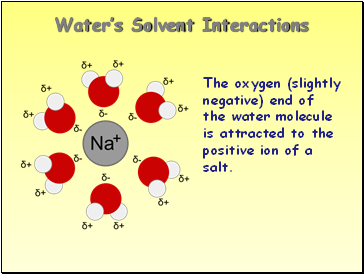
Water’s Solvent Interactions
The oxygen (slightly negative) end of the water molecule is attracted to the positive ion of a salt.
Slide 7
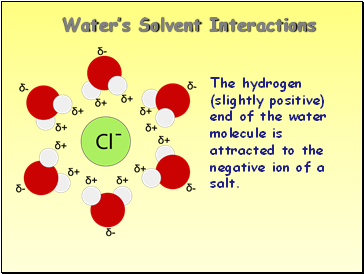
Water’s Solvent Interactions
The hydrogen (slightly positive) end of the water molecule is attracted to the negative ion of a salt.
Slide 8
Solubility Trends
The solubility of MOST solids increases with temperature.
The rate at which solids dissolve increases with increasing surface area of the solid.
The solubility of gases decreases with increases in temperature.
The solubility of gases increases with the pressure above the solution.
Slide 9
Therefore…
Solids tend to dissolve best when:
Heated
Stirred
Ground into small particles
Gases tend to dissolve best when:
The solution is cold
Pressure is high
Slide 10
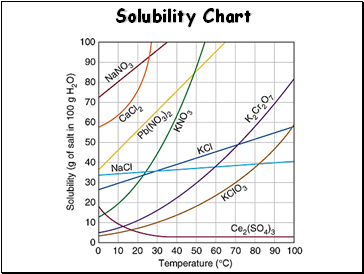
Solubility Chart
Slide 11
An electrolyte is:
A substance whose aqueous solution conducts an electric current.
A nonelectrolyte is:
A substance whose aqueous solution does not conduct an electric current.
Definition of Electrolytes and Nonelectrolytes
Slide 12
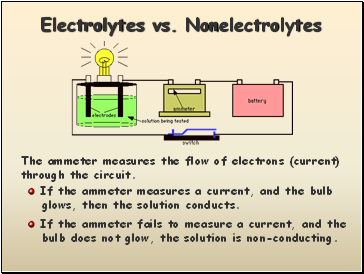
Electrolytes vs. Nonelectrolytes
The ammeter measures the flow of electrons (current)
through the circuit.
If the ammeter measures a current, and the bulb
glows, then the solution conducts.
1 2
Contents
- Classification of Matter
- Water’s Solvent Interactions
- Solubility Trends
- Therefore…
- Solubility Chart
- Electrolytes vs. Nonelectrolytes
- Answers to Electrolytes
- Colligative Properties
- Freezing Point Depression
Last added presentations
- Heat-Energy on the Move
- History of Modern Astronomy
- Motion
- Understanding Heat Transfer, Conduction, Convection and Radiation
- Practical Applications of Solar Energy
- Ch 9 Nuclear Radiation
- Newton’s laws of motion