Group 7, The HalogensPage
1
1
Slide 1
Group 7, the Halogens
Slide 2
Group 17—The Halogen Group
All the elements in Group 17 are nonmetals except for astatine, which is a radioactive metalloid.
Representative Elements
2
These elements are called halogens, which means “salt-former.”
All of the halogens form salts with sodium and with the other alkali metals.
Slide 3
Group 17—The Halogen Group
The halogen fluorine is the most reactive of the halogens in combining with other elements.
Representative Elements
Chlorine is less reactive than fluorine, and bromine is less reactive than chlorine.
Iodine is the least reactive of the four nonmetals.
Slide 4
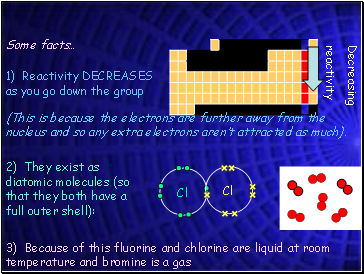
Some facts…
1) Reactivity DECREASES as you go down the group
(This is because the electrons are further away from the nucleus and so any extra electrons aren’t attracted as much).
2) They exist as diatomic molecules (so that they both have a full outer shell):
3) Because of this fluorine and chlorine are liquid at room temperature and bromine is a gas
Slide 5
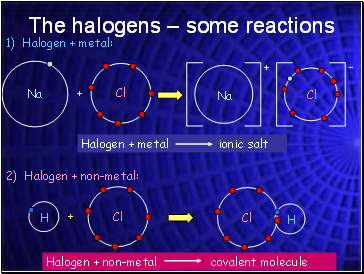
The halogens – some reactions
1) Halogen + metal:
+
2) Halogen + non-metal:
+
Slide 6
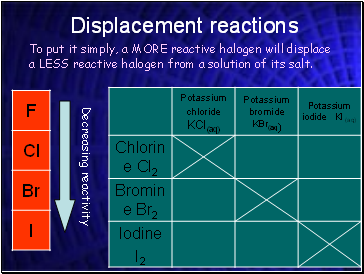
Displacement reactions
To put it simply, a MORE reactive halogen will displace a LESS reactive halogen from a solution of its salt.
Slide 7
Halogen compounds
Silver halides (e.g. silver chloride, silver bromide etc)
These are used in photographic paper. They are reduced by light and x-ray radiation to leave a silver photographic image.
Hydrogen halides (e.g. hydrogen chloride, hydrogen fluoride)
When these dissolve in water they make acids and will turn universal indicator red.
Slide 8
Section Check
Question 1
What does the term “halogen” mean?
Slide 9
Section Check
Answer
Halogen means “salt-former.” All the halogens form salts with sodium (and other alkali metals).
Contents
- Group 17—The Halogen Group
- Some facts…
- The halogens – some reactions
- Displacement reactions
- Halogen compounds
Last added presentations
- Resource Acquisition and Transport in Vascular Plants
- Ch 9 Nuclear Radiation
- Gravitation
- Geophysical Concepts, Applications and Limitations
- Solar Energy
- Newton’s laws of motion
- Radiation Safety and Operations