Bonding, Molecular Shape & StructurePage
1
1
Slide 1
Bonding, Molecular Shape & Structure
By
Dr. Fawaz Aldabbagh
Slide 2
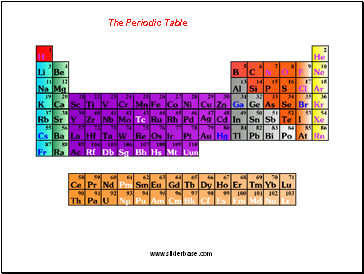
The Periodic Table
Slide 3
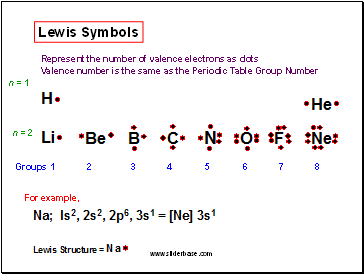
Lewis Symbols
Represent the number of valence electrons as dots
Valence number is the same as the Periodic Table Group Number
For example,
Groups 1 2 3 4 5 6 7 8
n = 1
n = 2
Slide 4
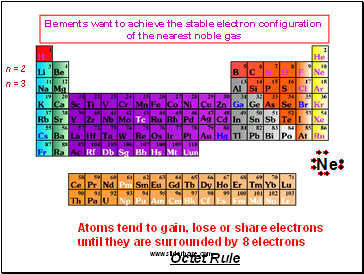
Elements want to achieve the stable electron configuration
of the nearest noble gas
Atoms tend to gain, lose or share electrons until they are surrounded by 8 electrons
Octet Rule
n = 2
n = 3
Slide 5
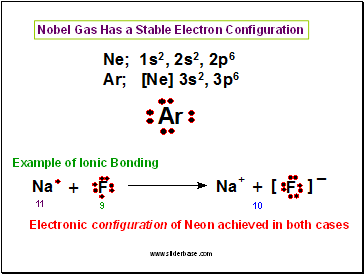
Nobel Gas Has a Stable Electron Configuration
Electronic configuration of Neon achieved in both cases
Example of Ionic Bonding
10
11
9
Slide 6
Ionic Bonding refers to electrostatic forces between ions, usually a metal cation and a non-metal anion
Covalent Bonding results from the sharing of two electrons between two atoms (usually non-metals) resulting in molecules
There are two types of bonding;
Octet Rule applies
Each Covalent Bond contains two electrons
Triple bond
Slide 7
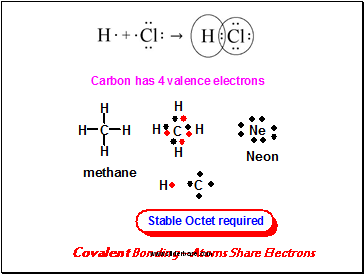
Covalent Bonding – Atoms Share Electrons
Slide 8
Hydrogen molecule, H2
Concentration of negative charge between two nuclei occurs in a covalent bond
7A elements (e.g. F) have one valence electron for covalent bonding, so to achieve octet
6A elements (e.g. O) use two valence electrons for covalent bonding, so to achieve octet
5A elements (e.g. N) use three valence electrons for covalent bonding, so to achieve octet
4A elements (e.g. C) use four valence electrons for covalent bonding, so to achieve octet
Slide 9
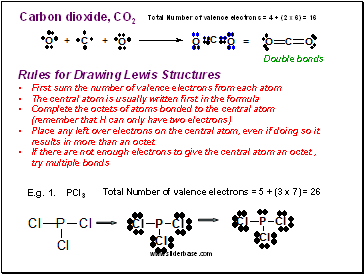
Carbon dioxide, CO2
Double bonds
Rules for Drawing Lewis Structures
First sum the number of valence electrons from each atom
The central atom is usually written first in the formula
Complete the octets of atoms bonded to the central atom (remember that H can only have two electrons)
Place any left over electrons on the central atom, even if doing so it results in more than an octet
If there are not enough electrons to give the central atom an octet , try multiple bonds
E.g. 1. PCl3
Contents
- Lewis Symbols
- Pauling scale of electronegativity;
- Electronegativity is dictated by
- Shapes of Molecules
- Trigonal Pyramidal
- Valence-Shell Electron-Pair Repulsion Theory (VSEPR)
- Valence Shell Electron-Pair Repulsion Theory (VSEPR)
- Molecules with Expanded Valence Shells
- Hydrogen Bonding & Water
- Dipole-dipole Attractive Forces
Last added presentations
- Solar Thermal Energy
- Space Radiation
- Gravitation
- Newton’s Laws of Motion
- Friction
- Newton’s laws of motion
- Mechanics Lecture