RadiationPage
2
2
Slide 15
Slide 16
Slide 17
Slide 18
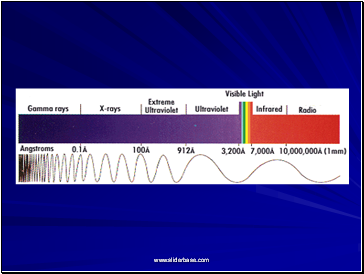
Types of Radiation
Radiation is classified into:
Ionizing radiation
Non-ionizing radiation
Slide 19
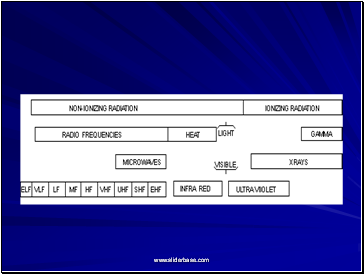
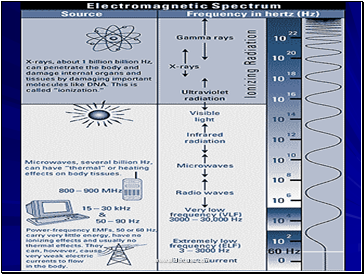
Ionizing Versus Non-ionizing Radiation
Ionizing Radiation
Higher energy electromagnetic waves (gamma) or heavy particles (beta and alpha).
High enough energy to pull electron from orbit.
Non-ionizing Radiation
Lower energy electromagnetic waves.
Not enough energy to pull electron from orbit, but can excite the electron.
Slide 20
Ionizing Radiation
Definition:
“ It is a type of radiation that is able to disrupt atoms and molecules on which they pass through, giving rise to ions and free radicals”.
Slide 21
Another Definition
Ionizing radiation
A radiation is said to be ionizing when it has enough energy to eject one or more electrons from the atoms or molecules in the irradiated medium. This is the case of a and b radiations, as well as of electromagnetic radiations such as gamma radiations, X-rays and some ultra-violet rays. Visible or infrared light are not, nor are microwaves or radio waves.
Slide 22
Primary Types of Ionizing Radiation
Alpha particles
Beta particles
Gamma rays (or photons)
X-Rays (or photons)
Neutrons
Slide 23
Alpha Particles: 2 neutrons and 2 protons
They travel short distances, have large mass
Only a hazard when inhaled Types and Characteristics of Ionizing Radiation Alpha Particles
Slide 24
Alpha Particles (or Alpha Radiation): Helium nucleus (2 neutrons and 2 protons); +2 charge; heavy (4 AMU). Typical Energy = 4-8 MeV; Limited range (<10cm in air; 60µm in tissue); High LET (QF=20) causing heavy damage (4K-9K ion pairs/µm in tissue). Easily shielded (e.g., paper, skin) so an internal radiation hazard. Eventually lose too much energy to ionize; become He.
Slide 25
Beta Particles
Beta Particles: Electrons or positrons having small mass and variable energy. Electrons form when a neutron transforms into a proton and an electron or:
Contents
- Definition of Radiation
- Radioactivity: Elements & Atoms
- Radioactivity
- Ionization
- Types of Radiation
- Ionizing Versus Non-ionizing Radiation
- Ionizing Radiation
- Another Definition
- Beta Particles
- Gamma Rays
- X-Rays
- Neutrons
- Exposure Limits
- Community Emergency Radiation
- Non-ionizing Radiation
- Effects
Last added presentations
- Mechanical, Electromagnetic, Electrical, Chemical and Thermal
- Health Physics
- Motion
- Ch 9 Nuclear Radiation
- Soil and Plant Nutrition
- Resource Acquisition and Transport in Vascular Plants
- Friction