Electron Quantum NumbersPage
1
1
Slide 1
Electron Orbitals
Cartoon courtesy of lab-initio.com
Slide 2
Quantum Mechanical Model of the Atom
Mathematical laws can identify the regions outside of the nucleus where electrons are most likely to be found.
These laws are beyond the scope of this class…
Slide 3
Heisenberg Uncertainty Principle
The more certain you are about where the electron is, the less certain you can be about where it is going.
The more certain you are about where the electron is going, the less certain you can be about where it is.
“One cannot simultaneously determine both the position and momentum of an electron.”
Werner
Heisenberg
Slide 4
Quantum Numbers
Each electron in an atom has a unique set of 4 quantum numbers which describe it.
Principal quantum number
Angular momentum quantum number
Magnetic quantum number
Spin quantum number
Slide 5
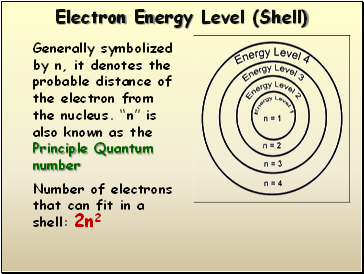
Electron Energy Level (Shell)
Generally symbolized by n, it denotes the probable distance of the electron from the nucleus. “n” is also known as the Principle Quantum number
Number of electrons that can fit in a shell:
2n2
Slide 6
Electron Orbitals
Orbital shapes are defined as the surface that contains 90% of the total electron probability.
An orbital is a region within an energy level where there is a probability of finding an electron.
The angular momentum quantum number, generally symbolized by l, denotes the orbital (subshell) in which the electron is located.
Slide 7
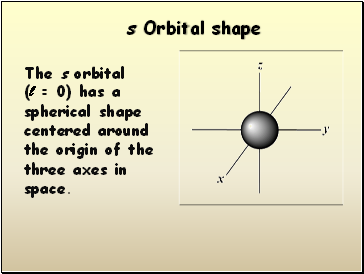
Orbital shape
The s orbital
(l = 0) has a spherical shape centered around
the origin of the three axes in space.
s
Slide 8
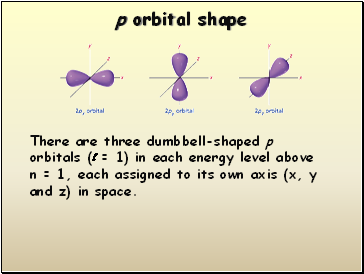
There are three dumbbell-shaped p orbitals (l = 1) in each energy level above n = 1, each assigned to its own axis (x, y and z) in space.
p orbital shape
Slide 9
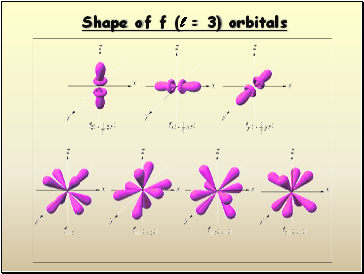
Things get a bit more complicated with the five d orbitals (l = 2) that are found in the d sublevels beginning with n = 3. To remember the shapes, think of “double dumbells”
…and a “dumbell
with a donut”!
d orbital shapes
Slide 10
1 2
Contents
- Electron Orbitals
- Quantum Mechanical Model of the Atom
- Heisenberg Uncertainty Principle
- Quantum Numbers
- Electron Energy Level (Shell)
- Electron Orbitals
- Orbital shape
- Energy Levels, Sublevels, Electrons
- Magnetic Quantum Number
- Orbital filling table
- Electron Spin
- Pauli Exclusion Principle
- Exclusion Warning!
- Assigning the Numbers
Last added presentations
- Upcoming Classes
- Gravitation
- Mechanics Lecture
- Soil and Plant Nutrition
- Newton’s law of universal gravitation
- Ch 9 Nuclear Radiation
- Magnetic field uses sound waves to ignite sun's ring of fire