Lab tests, results and sulphuric acidPage
2
2
Slide 11
Thermal decomposition
A “thermal decomposition” reaction occurs when a compound breaks down (“decomposition”) through the action of heat.
Practical work:
Perform a thermal decomposition reaction on each of these compounds and state:
The colour changes you observed
The reaction that happened
Slide 12
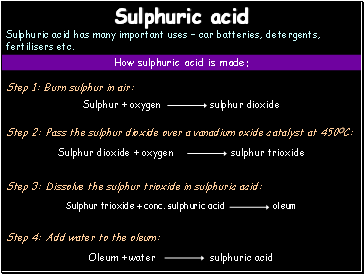
Sulphuric acid
Sulphuric acid has many important uses – car batteries, detergents, fertilisers etc.
How sulphuric acid is made:
Step 1: Burn sulphur in air:
Step 2: Pass the sulphur dioxide over a vanadium oxide catalyst at 450OC:
Step 3: Dissolve the sulphur trioxide in sulphuric acid:
Step 4: Add water to the oleum:
Slide 13
Sulphuric acid
Step 2 in the manufacture of sulphuric acid is an example of a reversible reaction:
What would happen if the temperature was decreased?
Solution – use 450OC as a compromise
The reaction would favour the production of sulphur trioxide BUT the reaction would happen at a slower rate.
1 2
Contents
- Testing for carbon dioxide
- Adding acid to carbonates
- Flame tests
- Metal ions
- Metal ions and precipitates
- Testing for chloride and sulphate ions
- Ammonium, nitrate, bromide and iodide ions
- Thermal decomposition
- Sulphuric acid
Last added presentations
- History of Modern Astronomy
- Mechanics Lecture
- Newton’s Laws of Motion
- Thermal Energy
- Gravitation
- Resource Acquisition and Transport in Vascular Plants
- Sensory and Motor Mechanisms