Earth HistoryPage
6
6
(Effects of Reduction)
1% Reduction In O3 2% increase in UV-B
Skin sunburns, tans, Skin cancer
Absorbed by DNA DNA damage
Possible eye cataracts
Interferes with photosynthesis
Organisms in 1st 5metre of the Oceans at risk
( phytoplankton in particular )
Slide 43
Chlorofluorocarbons & Ozone
Destruction of the Ozone Layer discovered in 1970’s by CFC’s ( Chlorofluorocarbons)
First synthesized Swartz (1892)
Used as refrigerants 1928 (Midgely & Henne)
CCl4 + xHF CCl(4-x)Fx + HCl
(Aerosol Propellants & Air conditioners)
Slide 44
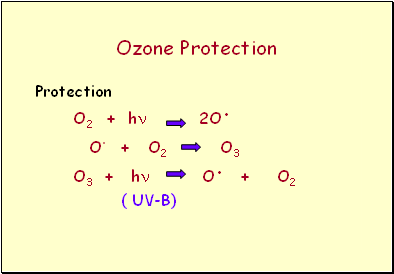
Ozone Protection
Protection
O2 + h 2O.
O. + O2 O3
O3 + h O. + O2
( UV-B)
Slide 45
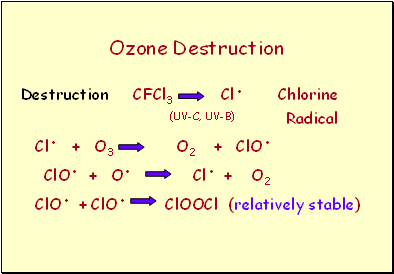
Ozone Destruction
Destruction CFCl3 Cl. Chlorine
(UV-C, UV-B) Radical
Cl. + O3 O2 + ClO.
ClO. + O. Cl. + O2
ClO. + ClO. ClOOCl (relatively stable)
Slide 46
Control of CFC’s
CFC’s are now under strict control and their use has been curtailed.
Australia signed the international treaty.
“The Montreal Protocol“ in June 1988 which has a program controlling the use and reduction of CFC’s.
Slide 47
Uses of CFC’s
Compound Use
CFC- 11 CFCl3 Refrigeration, aerosol, foam
CFC-12 CF2Cl2 sterilization, cosmetics
food freezing, pressurized
blowers.
CFC-113 CCl3CF3 solvent, cosmetics
Halon 1301 CBrF3 fire fighting (discontinued)
Slide 48
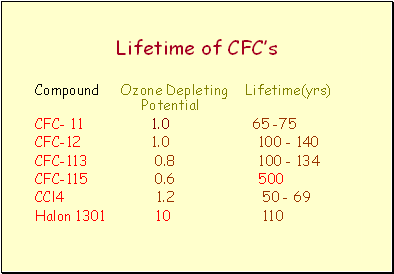
Lifetime of CFC’s
Compound Ozone Depleting Lifetime(yrs) Potential
CFC- 11 1.0 65 -75
CFC-12 1.0 100 - 140
CFC-113 0.8 100 - 134
CFC-115 0.6 500
CCl4 1.2 50 - 69
Halon 1301 10 110
Slide 49
Naming of CFC’s
( 90 Rule)
CFC’s name is related to its Formula.
CFC 123 123 + 90 = 213
The remaining bonds are allocated to Cl or Br
C = 2 , H =1 , F = 3 , Cl = ( 8 - 6) = 2
CFC 123 is CF3CHCl2
Letters with the number indicate an isomer.
C
H
F
Slide 50
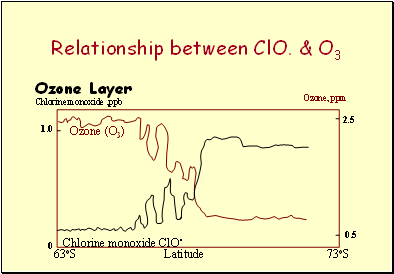
Chloromonoxide
Evidence for the destruction has been linked to the catalytically active Chloro monoxide ClO. & Ozone profiles as one goes South.
It is interesting to note how little Chloro monoxide effects the amounts of Ozone.
Slide 51
Relationship between ClO. & O3
Contents
- Atmospheric Chemistry
- Formation of the Earth
- Thermal Consequences
- Formation of the Mantle
- Isotope Distribution of the Earth
- Appearance of the Atmosphere
- Isotopes of Xe
- Distribution of Xe isotopes
- Differentiation
- Age of differentiation
- Ratios of Isotopes
- Conclusions from Isotope Analysis
- Collecting the evidence
- Early Atmosphere
- Origin of Life
- Formation of Simple Amino Acids
- Murchison Meteor
- Early Energy System
- Role of Blue Green Algae
- Decline of Anaerobic Bacteria
- Oxygen Rich Planet
- The trouble with oxygen
- The present atmosphere
- Distance from the Sun
- Influence of Earth’s Mass
- Escape Velocity
- No H or He in Earth’s Atmosphere
- Little CO2 in atmosphere
- Earth ,Venus & Mars
- Distribution of Gases on Earth Venus & Mars
- Role of Shellfish
- Triple point of H2O
- Water ( Solid,Liquid, Gas)
- Super Greenhouse & Acid Rain
- Current Atmosphere
- Present Level of Oxygen
- Structure of Atmosphere
- Ozone Layer
- Ozone and Radiation
- Effects of Reduction in Ozone
- Chlorofluorocarbons & Ozone
- Ozone Protection
- Ozone Destruction
- Control of CFC’s
- Uses of CFC’s
- Lifetime of CFC’s
- Naming of CFC’s
- Chloromonoxide
- Relationship between ClO. & O3
- Thickness of Ozone Layer
- Other Ozone Depleters
- Interactive Catalytic Forms
- Origin of Ozone Hole
- Ice crystal formation
- Possible Role of CO2
- Impenetrable Vortex formation
- PSC’s
- HCL attachment
- Role of ClONO2
- Formation of Cl. Radicals
- Hole Closure
- Dimer ClOOCl
- Antarctic and Arctic Vortexes
- Possible Link
- Further Reading
Last added presentations
- The Effects of Radiation on Living Things
- Motion
- Buoyancy
- Newton’s laws of motion
- Thermal Energy
- Static and Kinetic Friction
- Ch 9 Nuclear Radiation